| Citation: | Xia Liu, Zhenyu Zhang, Jie Liu, Xiang Gao, Bilan Wang. Research on α-mangostin nanoformulations for anti-ovarian cancer[J]. Materials Lab, 2025, 4(3): 250003. doi: 10.54227/mlab.20250003 |
Research on α-mangostin nanoformulations for anti-ovarian cancer
-
Abstract
Ovarian cancer is a type of malignant tumor with a high incidence rate, poor prognosis, and a tendency to relapse. There is a lack of effective therapeutic drugs in the treatment process of ovarian cancer. Alpha-mangostin, an oxyanthenone-type compound, has anti-tumor biological activities. Compared with traditional chemotherapeutic drugs, the advantage of natural source chemotherapeutic drugs lies in lower toxicity, killing tumor cells while reducing damage to normal cells, alleviating the adverse reactions and multi-drug resistance caused by chemotherapy. The nanoformulation was prepared using the thin-film dispersion method, with a particle size of approximately 60 nm. Compared to other drug formulations, its advantage lies in its good biocompatibility. Hydrogels are excellent drug carriers, possessing temperature sensitivity that enables controlled drug release. In this study, the water gel was used as a carrier to wrap the alpha-mangostin nanoparticles to form a nanocomposite hydrogel, and its effects on ovarian cancer cells were evaluated comprehensively, including cell proliferation, apoptosis, and cytotoxicity. Research has confirmed that alpha-mangostin nano-hydrogels can induce apoptosis and inhibit cell proliferation, showing good therapeutic effects on ovarian cancer cells. In summary, alpha-mangostin hydrogel has the potential to become a novel drug formulation for the treatment of ovarian cancer.
-
Keywords:
- Alpha-mangostin /
- apoptosis /
- hydrogel /
- ovarian cancer /
- proliferation
-
-
References
1. P. A. Konstantinopoulos, U. A. Matulonis, Nat. Cancer, 2023, 4, 1239. 2. E. A. Eisenhauer, Ann. Oncol., 2017, 28, viii61. 3. D. K. Armstrong, R. D. Alvarez, J. N. Bakkum-Gamez, L. Barroilhet, K. Behbakht, A. Berchuck, L. -M. Chen, M. Cristea, M. DeRosa, E. L. Eisenhauer, D. M. Gershenson, H. J. Gray, R. Grisham, A. Hakam, A. Jain, A. Karam, G. E. Konecny, C. A. Leath, J. Liu, H. Mahdi, L. Martin, D. Matei, M. McHale, K. McLean, D. S. Miller, D. M. O’Malley, S. Percac-Lima, E. Ratner, S. W. Remmenga, R. Vargas, T. L. Werner, E. Zsiros, J. L. Burns, A. M. Engh, J. Natl. Compr. Canc Netw., 2022, 20, 972 4. L. Kuroki, S. R. Guntupalli, BMJ, 2020, 371, m3773. 5. R. M. van Stein, A. G. J. Aalbers, G. S. Sonke, W. J. van Driel, JAMA Oncol., 2021, 7, 1231. 6. E. Ghisoni, M. Morotti, A. Sarivalasis, A. J. Grimm, L. Kandalaft, D. D. Laniti, G. Coukos, Nat. Rev. Clin. Oncol., 2024, 21, 801. 7. O. B. Garbuzenko, J. Sapiezynski, E. Girda, L. Rodriguez-Rodriguez, T. Minko, Small, 2024, 20, e2307462. 8. D. L. Richardson, R. N. Eskander, D. M. O’Malley, JAMA Oncol., 2023, 9, 851. 9. J. Kopecka, P. Trouillas, A. Č. Gašparović, E. Gazzano, Y. G. Assaraf, C, Drug Resist. Updat., 2020, 49, 100670. 10. Q. Gao, J. Feng, W. Liu, C. Wen, Y. Wu, Q. Liao, L. Zou, X. Sui, T. Xie, J. Zhang, Y. Hu, Adv. Drug. Deliv Rev., 2022, 188, 114445. 11. T. Sathasivam, J. Kai, S. Sugiarto, Y. Yu, D. X. Y. Soo, Q. Zhu, J. Merzaban, D. Kai, Adv. Healthc. Mater., 2023, 12, e2300024. 12. Y. Liang, Z.-Y. Liu, P. -Y. Wang, Y.-J. Li, R.-R. Wang, S.-Y. Xie, J. Control. Release, 2021, 336, 396 13. L. Wang, X. Wang, X. Zhu, L. Zhong, Q. Jiang, Y. Wang, Q. Tang, Q. Li, C. Zhang, H. Wang, D. Zou, Mol. Cancer, 2024, 23, 66. 14. D. R. Freyer, P. R. Brock, K. W. Chang, L. L. Dupuis, S. Epelman, K. Knight, D. Mills, R. Phillips, E. Potter, D. Risby, P. Simpkin, M. Sullivan, S. Cabral, P. D. Robinson, L. Sung, Lancet Child Adolesc. Health, 2020, 4, 141. 15. M. C. Nauman, J. J. Johnson, Pharmacol. Res., 2022, 175, 106032. 16. W. M. Aizat, F. H. Ahmad-Hashim, S. N. Syed Jaafar, J. Adv. Res., 2019, 20, 61. 17. I. Ray-Coquard, P. Pautier, S. Pignata, D. Pérol, A. González-Martín, R. Berger, K. Fujiwara, I. Vergote, N. Colombo, J. Mäenpää, F. Selle, J. Sehouli, D. Lorusso, E. M. Guerra Alía, A. Reinthaller, S. Nagao, C. Lefeuvre-Plesse, U. Canzler, G. Scambia, A. Lortholary, F. Marmé, P. Combe, N. de Gregorio, M. Rodrigues, P. Buderath, C. Dubot, A. Burges, B. You, E. Pujade-Lauraine, P. Harter, N. Engl. J. Med., 2019, 381, 2416. 18. G. A. Mohamed, A. M. Al-Abd, A. M. El-Halawany, H. M. Abdallah, S. R. M. Ibrahim, J. Ethnopharmacol., 2017, 198, 302. 19. A. M. Gutierrez, E. M. Frazar, M. V. X Klaus, P. Paul, J. Z. Hilt, Adv. Healthc. Mater., 2022, 11, e2101820. 20. A. S. Mikhail, R. Morhard, M. Mauda-Havakuk, M. Kassin, A. Arrichiello, B. J. Wood, Adv. Drug Deliv. Rev., 2023, 202, 115083. 21. R. Liu, Q. Liang, J. -Q. Luo, Y. -X. Li, X. Zhang, K. Fan, J. -Z. Du, Adv. Sci., 2024, 11, e2305217 22. Y. Han, P. Wen, J. Li, K. Kataoka, J. Control. Release, 2022, 345, 709. 23. K. N. Moore, A. M. Oza, N. Colombo, A. Oaknin, G. Scambia, D. Lorusso, G. E. Konecny, S. Banerjee, C. G. Murphy, J. L. Tanyi, H. Hirte, J. A. Konner, P. C. Lim, M. Prasad-Hayes, B. J. Monk, P. Pautier, J. Wang, A. Berkenblit, I. Vergote, M. J. Birrer, Ann. Oncol., 2021, 32, 757. 24. M. Ashrafizadeh, A. Zarrabi, A. Bigham, A. Taheriazam, Y. Saghari, S. Mirzaei, M. Hashemi, K. Hushmandi, H. Karimi-Maleh, E. Nazarzadeh Zare, E. Sharifi, Y. N. Ertas, N. Rabiee, G. Sethi, M. Shen, Med. Res. Rev., 2023, 43, 2115-2176. 25. Y. Hua, H. Yin, X. Liu, J. Xie, W. Zhan, G. Liang, Y. Shen, Adv. Sci. , 2022, 9, e2202260. 26. C. Sessa, D. T. Schneider, F. Planchamp, K. Baust, E. I. Braicu, N. Concin, J. Godzinski, W. G. McCluggage, D. Orbach, P. Pautier, F. A. Peccatori, P. Morice, G. Calaminus, Lancet Oncol., 2020, 21, e360. 27. D. M. O’Malley, J. Natl. Compr. Canc. Netw., 2019, 17, 619. 28. S. Zhang, Y. Zhang, W. Wang, Y. Hu, X. Chen, B. Wang, X. Gao, CCL, 2024, 35, 1096. 29. S. Zuo, J. Song, J. Zhang, Z. He, B. Sun, J. Sun, Theranostics, 2021, 11, 7471. 30. Y. Xin, M. Huang, W. W. Guo, Q. Huang, L. Z. Zhang, G. Jiang, Mol. Cancer, 2017, 16, 134. 31. Y. Yang, J. Guo, L. Huang, Trends Pharmacol. Sci., 2020, 41, 701. 32. R. Zhong, S. Talebian, B. B. Mendes, G. Wallace, R. Langer, J. Conde, J. Shi, Nat. Mater., 2023, 22, 818. 33. W. Shen, P. Pei, C. Zhang, J. Li, X. Han, T. Liu, X. Shi, Z. Su, G. Han, L. Hu, K. Yang, ACS Nano, 2023, 17, 23998. 34. Y. Wu, X. Chang, G. Yang, L. Chen, Q. Wu, J. Gao, R. Tian, W. Mu, J. J. Gooding, X. Chen, S. Sun, Adv. Mater., 2023, 35, e2210787. 35. Y. Luo, J. Li, Y. Hu, F. Gao, G. Pak-Heng Leung, F. Geng, C. Fu, J. Zhang, Acta Pharm. Sin. B, 2020, 10, 2227. 36. X. Zhu, J. Li, H. Ning, Z. Yuan, Y. Zhong, S. Wu, J. -Z. Zeng, Front. Pharmacol., 2021, 12, 739658 37. T. Das, U. Anand, S. K. Pandey, C. R. Ashby, Y. G. Assaraf, Z. -S. Chen, A. Dey, Drug Resist. Updat., 2021, 55, 100754 38. C. Jia, Q. Liu, M. Zhang, C. Han, X. Luo, Y. Zhou, Y. Liu, L. Zhang, Small, 2025, 21, e2408095. 39. Y. You, J. Xu, Y. Liu, H. Li, L. Xie, C. Ma, Y. Sun, S. Tong, K. Liang, S. Zhou, F. Ma, Q. Song, W. Xiao, K. Fu, C. Dai, S. Li, J. Lei, Q. Mei, X. Gao, J. Chen, ACS Nano, 2023, 17, 8646. 40. F. I. Montalto, F. De Amicis, Cells, 2020, 9, 2648. -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
Figure 1.
Characterization of alpha-mangostin nanoformulations. a The zeta potential graph of the alpha-mangostin nanoformulation. b The particle size distribution graph of the alpha-mangostin nanoformulation. c The transmission electron microscopy (TEM) image of the alpha-mangostin nanoformulation. d The schematic diagram of the alpha-mangostin nanoformulation. e The process of F127 forming a thermosensitive gel.
-
Figure 2.
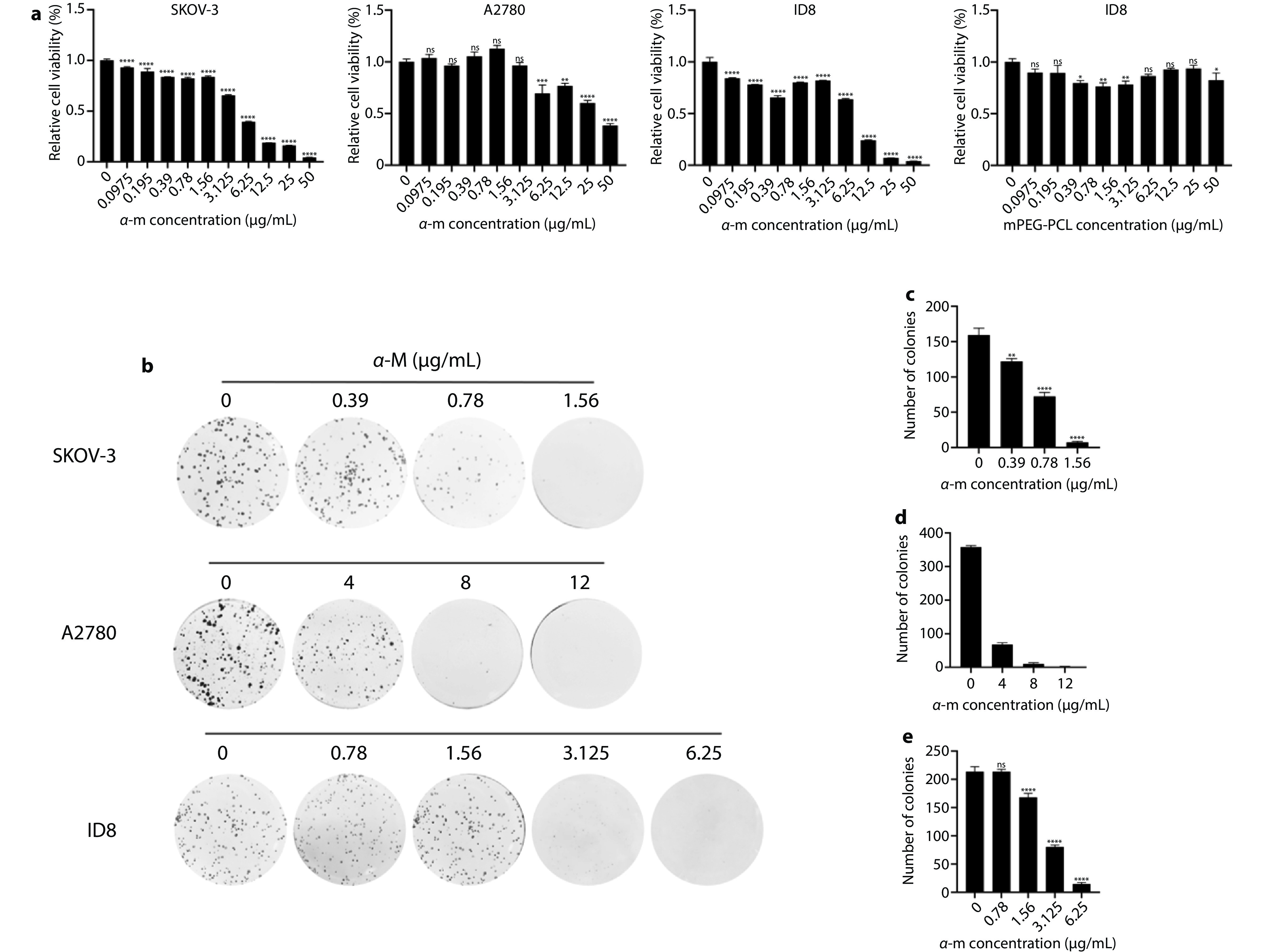
Cell viability studies and proliferation inhibition effects of alpha-mangostin. a After co-incubating different concentrations of alpha-mangostin nanoformulations with ovarian cancer cells SKOV-3, A2780, and ID8 for 24 or 48 hours in vitro, cell viability was assessed using the MTT assay kit (n=3). After co-incubating ovarian cancer cells ID8 with various concentrations of mPEG-PCL for 24 hours in vitro, cell viability was assessed using the MTT assay kit (n=3). b Two weeks after co-incubating different concentrations of alpha-mangostin nanoformulations with ovarian cancer cells SKOV-3, A2780, and ID8 in vitro, colony formation of cells in different treatment groups (n=3). c Count of clones formed by SKOV-3 cells after treatment with different concentrations of alpha-mangostin nanoformulations for two weeks. d Count of clones formed by A2780 cells after treatment with different concentrations of alpha-mangostin nanoformulations for two weeks. e Count of clones formed by ID8 cells after treatment with different concentrations of alpha-mangostin nanoformulations for two weeks.
-
Figure 3.
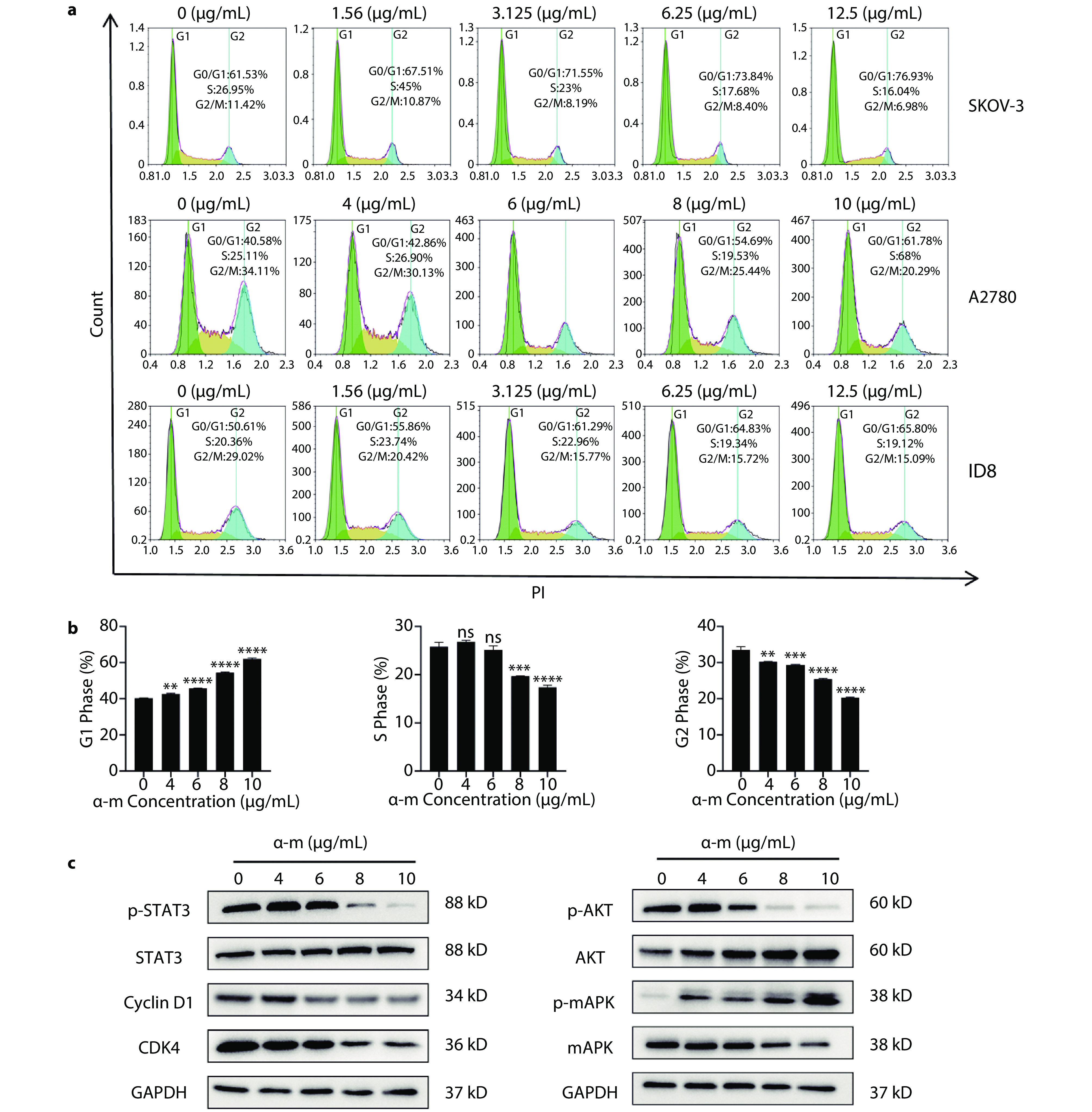
The cell cycle arrest effect of alpha-mangostin. a Determination of the cell cycle of ovarian cancer cell lines SKOV-3, A2780, and ID8 in vitro using alpha-mangostin nanoformulations. b The percentage of A2780 cells in the G1, S, and G2 phases of the cell cycle after treatment with alpha-mangostin nanoformulations. c Expression of proliferation and cell cycle-related proteins in ovarian cancer A2780 cells after treatment with various concentrations of alpha-mangostin nanoformulations for 48 hours (n=3).
-
Figure 4.
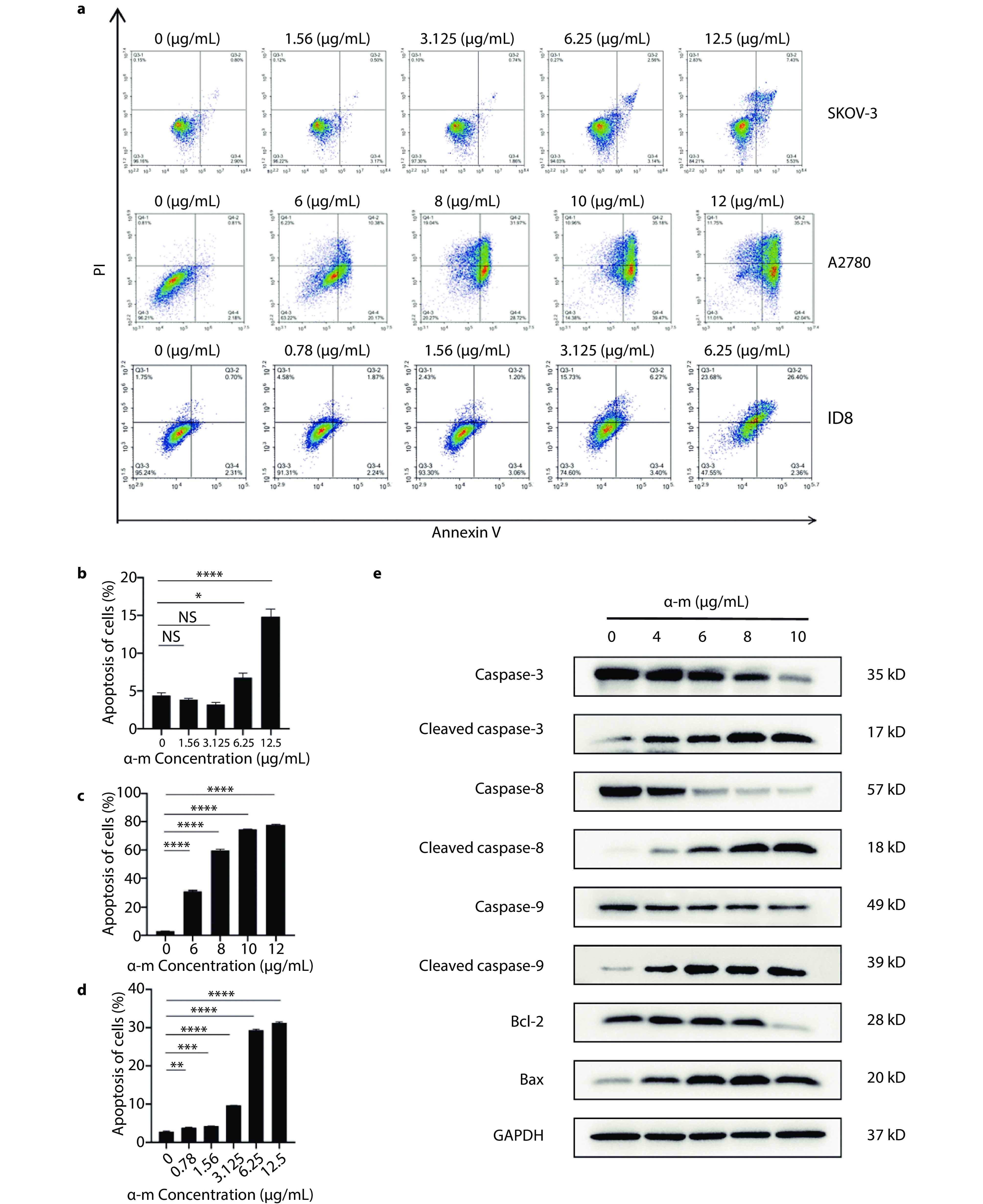
Determination of the apoptotic effect of alpha-mangostin. a Apoptosis assay of alpha-mangostin nanoformulations against ovarian cancer cell lines SKOV-3, A2780, and ID8 in vitro (n=3). b Apoptosis ratio of ovarian cancer SKOV-3 cells after treatment with various concentrations of alpha-mangostin nanoformulations for 48 hours. c Apoptosis ratio of ovarian cancer A2780 cells after treatment with various concentrations of alpha-mangostin nanoformulations for 48 hours. d Apoptosis ratio of ovarian cancer ID8 cells after treatment with various concentrations of alpha-mangostin nanoformulations for 48 hours. e Expression of apoptosis-related proteins in ovarian cancer A2780 cells after treatment with various concentrations of alpha-mangostin nanoformulations for 48 hours (n=3).
-
Figure 5.
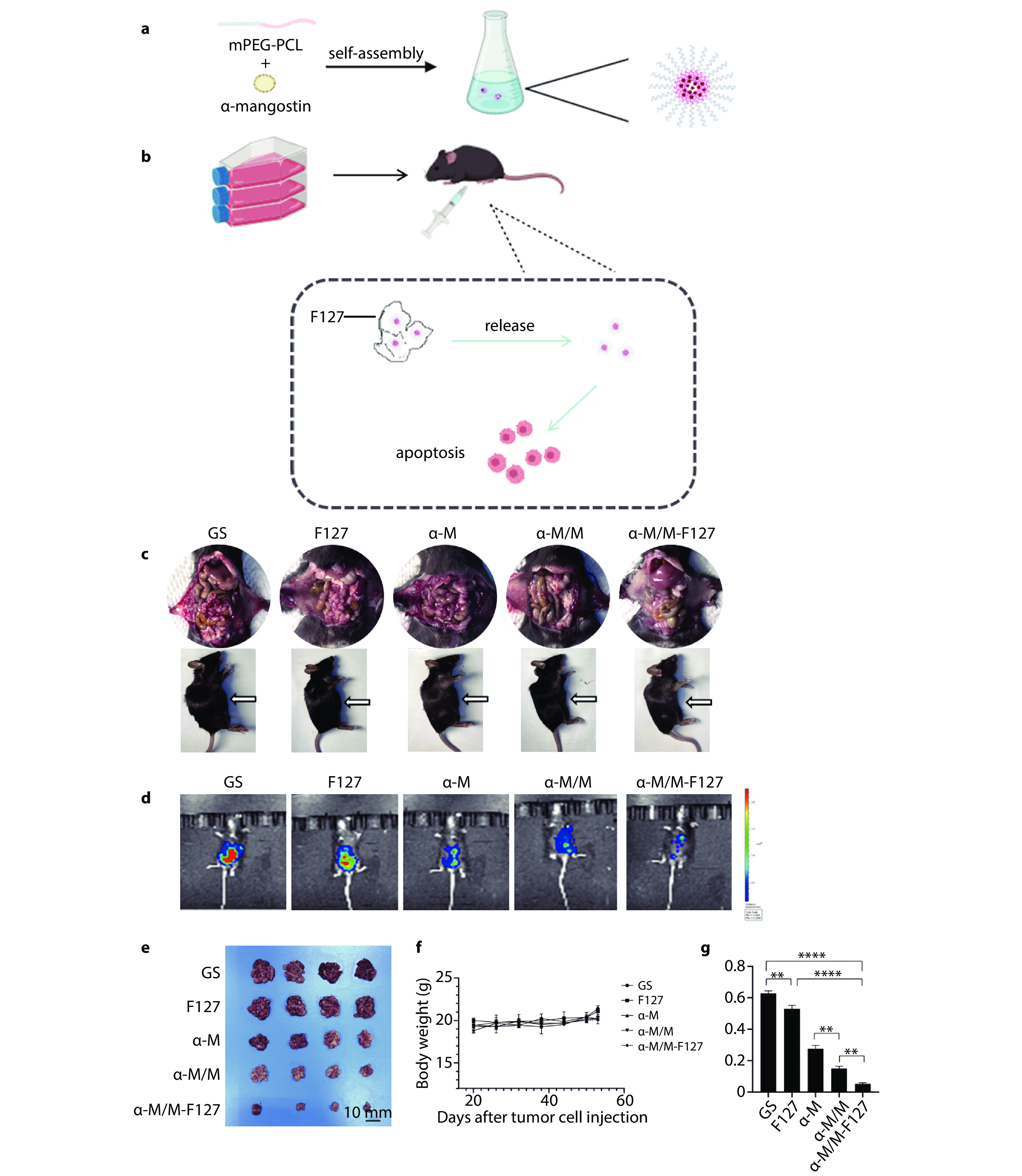
Schematic diagram of alpha-mangostin nanoformulation preparation and induction of tumor cell apoptosis, and the antitumor activity of F127 hydrogel-encapsulated alpha-mangostin nanoformulations in an ovarian cancer peritoneal dissemination model. a Schematic diagram of the preparation of alpha-mangostin nanoformulations. b Schematic diagram of the inoculation of ID8-luc ovarian cancer peritoneal animal model and the in vivo action of the drug. c Schematic diagram of the preparation of alpha-mangostin nanoformulations. d In vivo imaging images of tumor-bearing mice treated with different groups. e Images of tumors from different groups. f Graph of body weight changes in tumor-bearing mice. g Tumor weight (n=4).
-
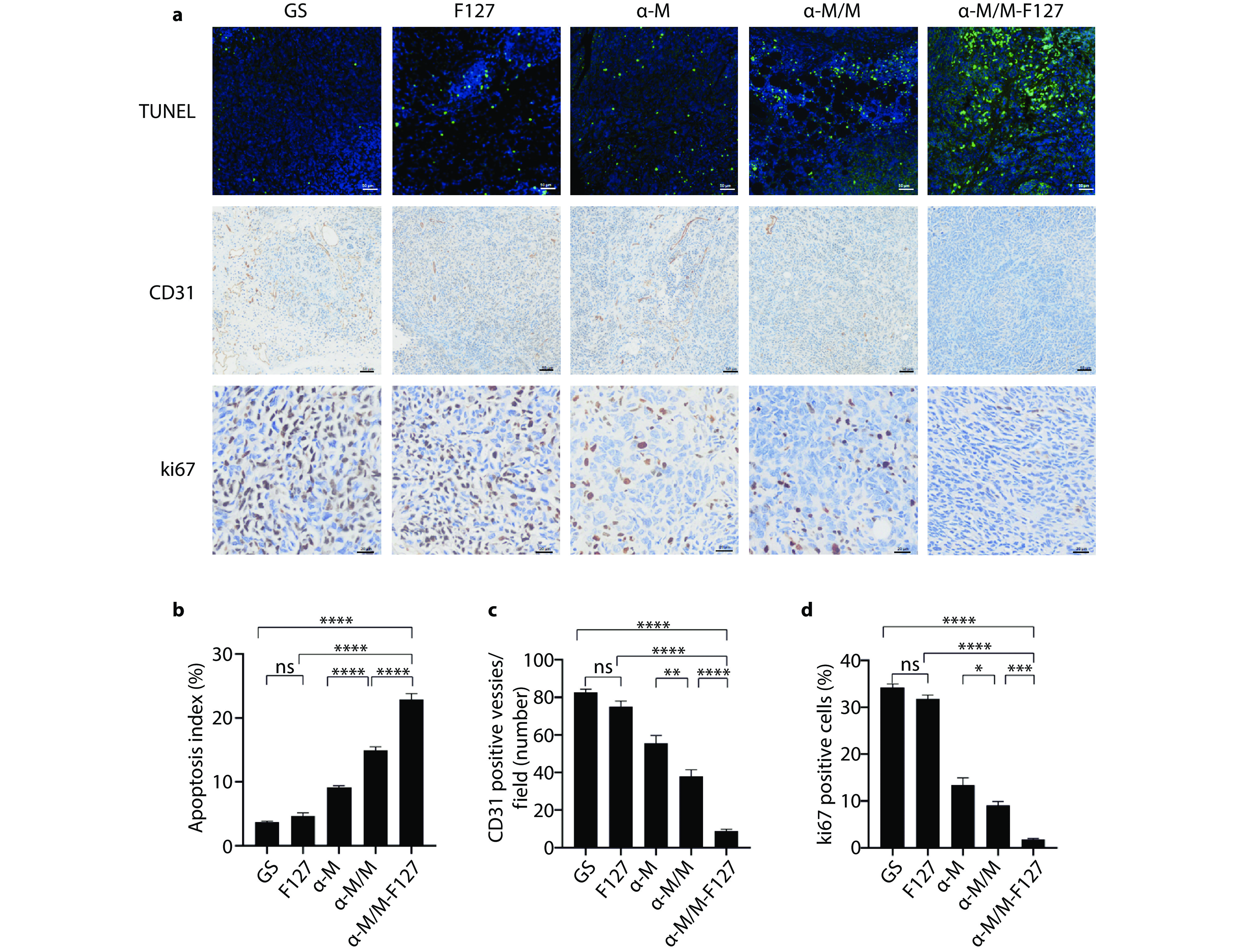
Figure 6.
In vivo determination of cell apoptosis, angiogenesis, and proliferation. a TUNEL, CD31, and Ki67 immunohistochemical staining in the mouse ID8 ovarian cancer model to assess the effects of different drug treatments on tumor cell apoptosis, angiogenesis, and in vivo proliferation. b Average percentage of TUNEL-positive cells (n=5). c Average number of positive blood vessels (n=5). d Average percentage of Ki67-positive cells (n=5).
-
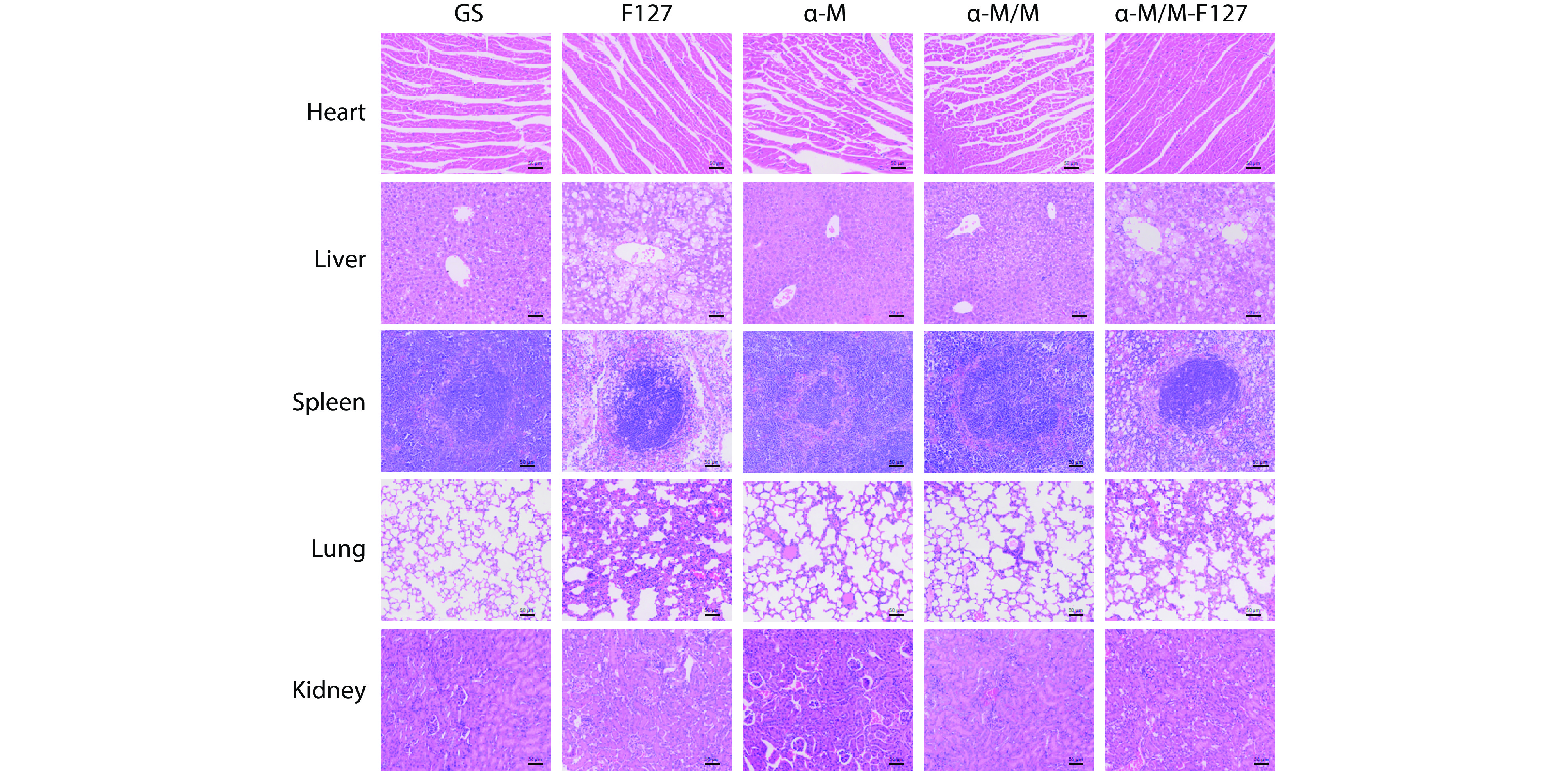
Figure 7.
Representative H&E staining images of hearts, livers, spleens, kidneys, and lungs from different groups at the end of the treatment period, (n=5).
-
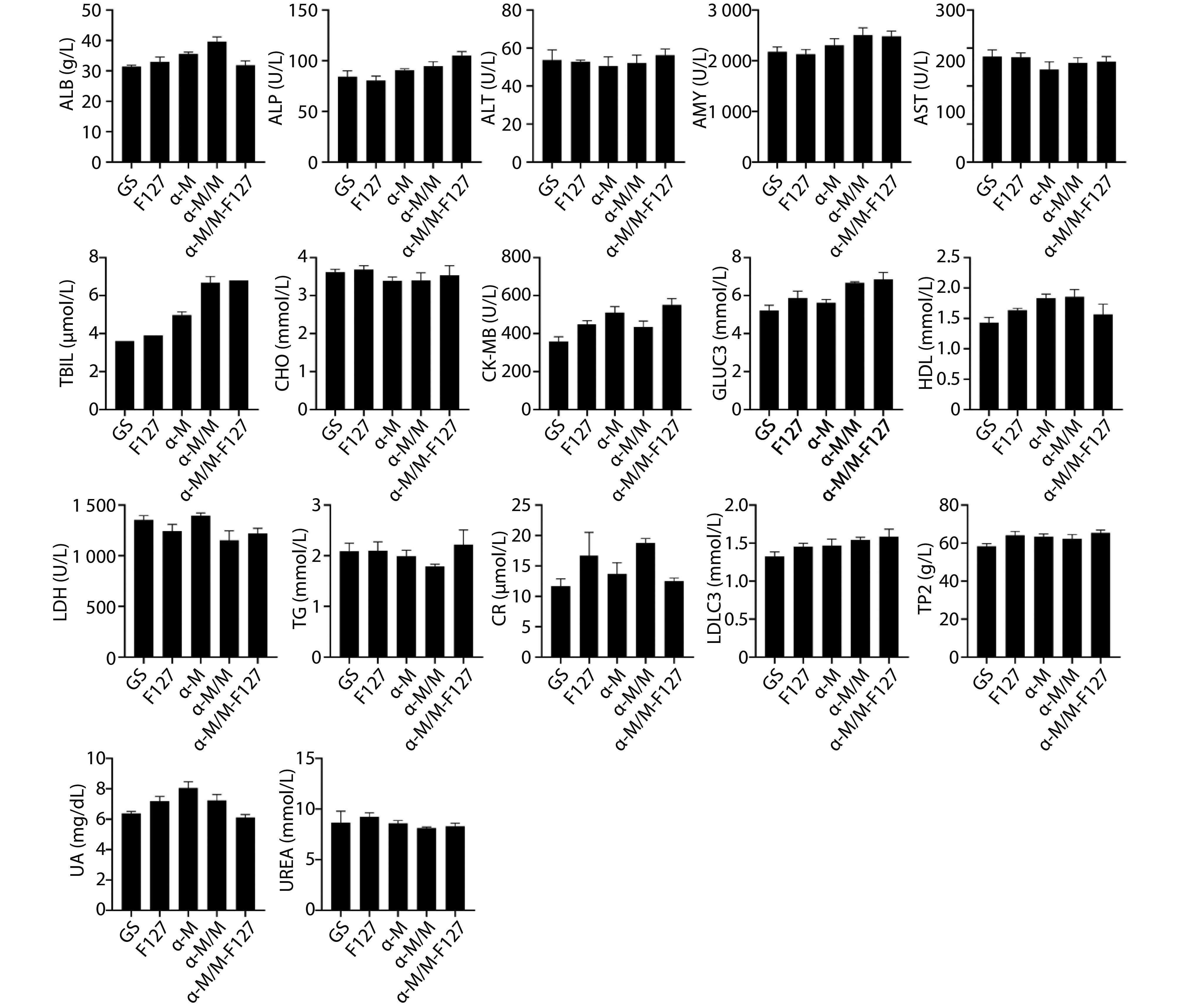
Figure 8.
Analysis of serum biochemical markers from different groups, (n=5).

 Xia Liu graduated from Hainan University in 2023 with a bachelor's degree and is currently a master's student at the National Key Laboratory of Biotherapy, Sichuan University. She has joined Professor Xiang Gao's research group, focusing on research areas such as anticancer drugs and nanomaterials.
Xia Liu graduated from Hainan University in 2023 with a bachelor's degree and is currently a master's student at the National Key Laboratory of Biotherapy, Sichuan University. She has joined Professor Xiang Gao's research group, focusing on research areas such as anticancer drugs and nanomaterials. 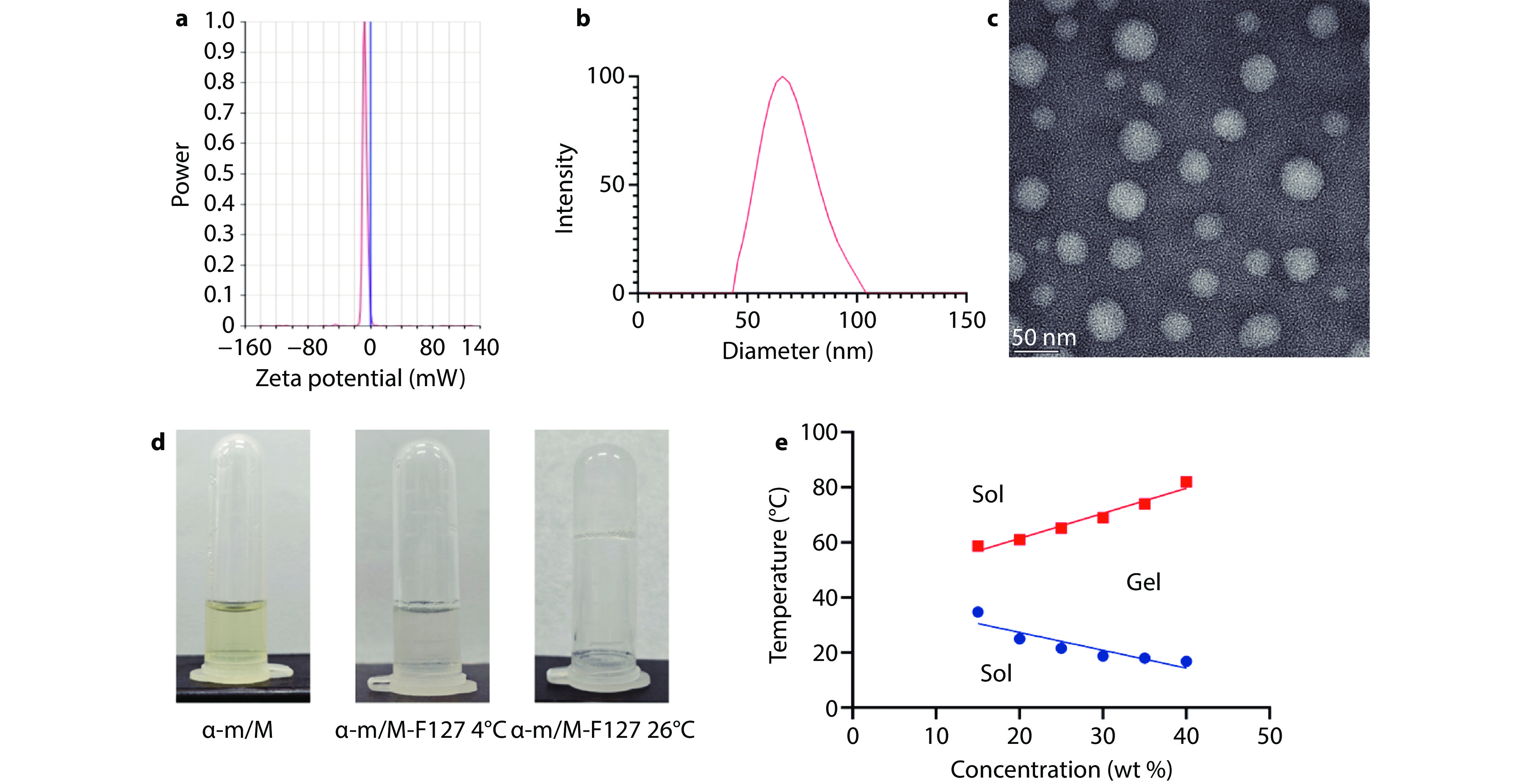

 DownLoad:
DownLoad: