| Citation: | Jinfeng Dong, Jing Gao, Qingyu Yan. High entropy strategy on thermoelectric materials[J]. Materials Lab, 2023, 2(2): 230001. doi: 10.54227/mlab.20230001 |
High entropy strategy on thermoelectric materials
-
Abstract
High-entropy materials, which consist of multiple elements occupying a single sublattice in a disordered manner, have emerged as innovative material systems with various promising applications. Many macroscopic physical properties, such as electrical transport and thermal transport, are closely related to the periodic distribution of atoms. In high-entropy compounds, the long-range periodic arrangement of atoms is broken down by the disordered distribution of various elements, which would lead to changes in physical properties. Therefore, the high-entropy idea will open new avenues for designing these functional materials with promising performance and high reliability. This perspective focuses on the high-entropy strategies of thermoelectric materials, discussing how high entropy will alter their properties. The possible routes of designing high-entropy high-performance thermoelectric materials are prospected, which can also provide enlightenment for the development of high-entropy systems in other research fields.
-
Keywords:
- high entropy compound /
- thermoelectric materials
-
-
References
1. J. W. Yeh, S. K. Chen, S. J. Lin, J. Y. Gan, T. S. Chin, T. T. Shun, C. H. Tsau, S. Y. Chang, Adv. Eng. Mater., 2004, 6, 299 2. E. P. George, D. Raabe, R. O. Ritchie, Nat. Rev. Mater., 2019, 4, 515 3. C. Oses, C. Toher, S. Curtarolo, Nat. Rev. Mater., 2020, 5, 295 4. B. Gludovatz, A. Hohenwarter, D. Catoor, E. H. Chang, E. P. George, R. O. Ritchie, Science, 2014, 345, 1153 5. B. Jiang, Y. Yu, J. Cui, X. Liu, L. Xie, J. Liao, Q. Zhang, Y. Huang, S. Ning, B. Jia, B. Zhu, S. Bai, L. Chen, S. J. Pennycook, J. He, Science, 2021, 371, 830 6. B. Yang, Y. Zhang, H. Pan, W. Si, Q. Zhang, Z. Shen, Y. Yu, S. Lan, F. Meng, Y. Liu, H. Huang, J. He, L. Gu, S. Zhang, L.-Q. Chen, J. Zhu, C.-W. Nan, Y.-H. Lin, Nat. Mater., 2022, 21, 1074 7. D. Nita, Materials Lab, 2022, 1, 220001 8. C. M. Rost, E. Sachet, T. Borman, A. Moballegh, E. C. Dickey, D. Hou, J. L. Jones, S. Curtarolo, J.-P. Maria, Nat. Commun., 2015, 6, 8485 9. D. B. Miracle, O. N. Senkov, Acta. Mater., 2017, 122, 448 10. Y. F. Ye, Q. Wang, J. Lu, C. T. Liu, Y. Yang, Mater. Today, 2016, 19, 349 11. B. Cantor, I. T. H. Chang, P. Knight, A. J. B. Vincent, Mater. Sci. Eng.: A, 2004, 375−377, 213 12. W. Huang, J. Zhang, D. Liu, W. Xu, Y. Wang, J. Yao, H. T. Tan, K. N. Dinh, C. Wu, M. Kuang, W. Fang, R. Dangol, L. Song, K. Zhou, C. Liu, J. W. Xu, B. Liu, Q. Yan, ACS Nano, 2020, 14, 17640 13. Y. Sun, S. Dai, Sci. Adv., 2021, 7, eabg1600 14. D. Bérardan, S. Franger, D. Dragoe, A. K. Meena, N. Dragoe, Phy. Status Solidi - Rapid Res. Lett., 2016, 10, 328 15. R. Liu, H. Chen, K. Zhao, Y. Qin, B. Jiang, T. Zhang, G. Sha, X. Shi, C. Uher, W. Zhang, L. Chen, Adv. Mater., 2017, 29, 1702712 16. Y. B. Luo, S. Q. Hao, S. T. Cai, T. J. Slade, Z. Z. Luo, V. P. Dravid, C. Wolverton, Q. Y. Yan, M. G. Kanatzidis, J. Am. Chem. Soc., 2020, 142, 15187 17. B. Jiang, Y. Yu, J. Cui, X. Liu, L. Xie, J. Liao, Q. Zhang, Y. Huang, S. Ning, B. Jia, B. Zhu, S. Bai, L. Chen, S. J. Pennycook, J. He, Science, 2021, 371, 830 18. Z. Chen, X. Zhang, Y. Pei, Adv. Mater., 2018, 30, 1705617 19. T.-R. Wei, M. Guan, J. Yu, T. Zhu, L. Chen, X. Shi, Joule, 2018, 2, 2183 20. G. J. Snyder, E. S. Toberer, Nat. Mater., 2008, 7, 105 21. J. Dong, Y. Jiang, Y. Sun, J. Liu, J. Pei, W. Li, X. Y. Tan, L. Hu, N. Jia, B. Xu, Q. Li, J.-F. Li, Q. Yan, M. G. Kanatzidis, J. Am. Chem. Soc., 2023, 145, 1988 22. Z. Chen, X. Zhang, S. Lin, L. Chen, Y. Pei, Natl. Sci. Rev., 2018, 5, 888 23. E. S. Toberer, A. Zevalkink, G. J. Snyder, J. Mater. Chem., 2011, 21, 15843 24. X. Wang, H. Yao, Z. Zhang, X. Li, C. Chen, L. Yin, K. Hu, Y. Yan, Z. Li, B. Yu, F. Cao, X. Liu, X. Lin, Q. Zhang, ACS Appl. Mater. Interfaces, 2021, 13, 18638 25. Z. Ma, T. Xu, W. Li, Y. Cheng, J. Li, D. Zhang, Q. Jiang, Y. Luo, J. Yang, Adv. Funct. Mater., 2021, 31, 2103197 26. B. Jiang, W. Wang, S. Liu, Y. Wang, C. Wang, Y. Chen, L. Xie, M. Huang, J. He, Science, 2022, 377, 208 27. S. Zhi, J. Li, L. Hu, J. Li, N. Li, H. Wu, F. Liu, C. Zhang, W. Ao, H. Xie, X. Zhao, S. J. Pennycook, T. Zhu, Adv. Sci., 2021, 8, 2100220 28. National Academies of Sciences, and Medicine, High-Entropy Materials, Ultra-Strong Molecules, and Nanoelectronics: Emerging Capabilities and Research Objectives: Proceedings of a Workshop, National Academies Press, America, 2020. 29. L. Hu, Y. Zhang, H. Wu, J. Li, Y. Li, M. McKenna, J. He, F. Liu, S. J. Pennycook, X. Zeng, Adv. Energy Mater., 2018, 8, 1802116 30. R. J. Korkosz, T. C. Chasapis, S.-h. Lo, J. W. Doak, Y. J. Kim, C.-I. Wu, E. Hatzikraniotis, T. P. Hogan, D. N. Seidman, C. Wolverton, V. P. Dravid, M. G. Kanatzidis, J. Am. Chem. Soc., 2014, 136, 3225 31. M. Lannoo, Point defects in semiconductors I: theoretical aspects, Springer Science & Business Media, Germany, 2012. 32. X. Y. Tan, J. Dong, N. Jia, H.-X. Zhang, R. Ji, A. Suwardi, Z.-L. Li, Q. Zhu, J.-W. Xu, Q.-Y. Yan, Rare Metals, 2022, 41, 3027 33. S. Roychowdhury, T. Ghosh, R. Arora, M. Samanta, L. Xie, N. K. Singh, A. Soni, J. He, U. V. Waghmare, K. Biswas, Science, 2021, 371, 722 34. C. M. Rost, Z. Rak, D. W. Brenner, J.-P. Maria, J. Am. Ceram. Soc., 2017, 100, 2732 35. W. Guo, W. Dmowski, J.-Y. Noh, P. Rack, P. K. Liaw, T. Egami, Metall. Mater. Trans. A, 2013, 44, 1994 36. J. Gao, W. Li, J. Liu, Q. Li, J.-F. Li, Research, 2022, 2022, 9782343 37. T. Ghosh, S. Roychowdhury, M. Dutta, K. Biswas, ACS Energy Lett., 2021, 6, 2825 38. H. Wang, X. Cao, Y. Takagiwa, G. J. Snyder, Mater. Horiz., 2015, 2, 323 39. Z. Rao, P.-Y. Tung, R. Xie, Y. Wei, H. Zhang, A. Ferrari, T. P. C. Klaver, F. Körmann, P. T. Sukumar, A. Kwiatkowski da Silva, Y. Chen, Z. Li, D. Ponge, J. Neugebauer, O. Gutfleisch, S. Bauer, D. Raabe, Science, 2022, 378, 78 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
Figure 1.
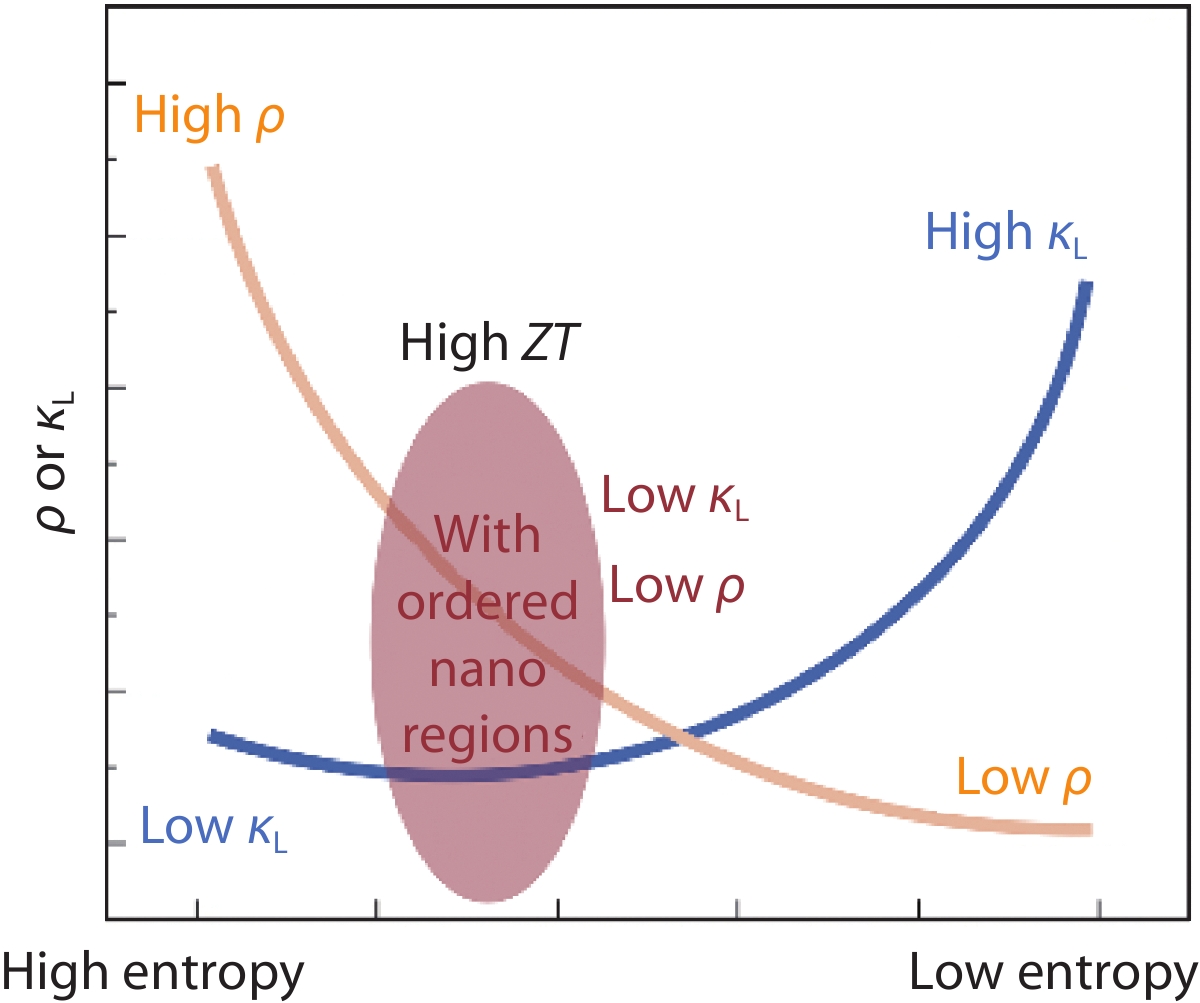
a High-entropy compound with a typical Fm-3m structure (AB, sublattice A is engineered with high-entropy). b High-entropy perovskite (ABO3, sublattice A is engineered with high entropy). c Schematic display of electrical resistivity and lattice thermal conductivity for the compound with different entropies.

 Jinfeng Dong is now a postdoctoral research fellow at Nanyang Technological University, Singapore. He received his Ph.D. degree from Tsinghua University, China, in 2020. His research focuses on thermoelectric semiconductors.
Jinfeng Dong is now a postdoctoral research fellow at Nanyang Technological University, Singapore. He received his Ph.D. degree from Tsinghua University, China, in 2020. His research focuses on thermoelectric semiconductors.  Jing Gao is now a postdoctoral research fellow at National University of Singapore, Singapore. She received her Ph.D. degree from Tsinghua University, China, in 2021. Her research focuses on dielectric materials for energy storge and neuromorphic computing.
Jing Gao is now a postdoctoral research fellow at National University of Singapore, Singapore. She received her Ph.D. degree from Tsinghua University, China, in 2021. Her research focuses on dielectric materials for energy storge and neuromorphic computing.  Qingyu Yan is currently a professor in Nanyang Technology University. He finished his Ph.D. degree from the State University of New York at Stony Brook. After that, he joined Rensselaer Polytechnic Institute as a research fellow. He joined Nanyang Technological University as an Assistant Professor in early 2008. Yan’s research interests mainly include advanced electrode materials for energy storage devices and high-efficiency thermoelectrics.
Qingyu Yan is currently a professor in Nanyang Technology University. He finished his Ph.D. degree from the State University of New York at Stony Brook. After that, he joined Rensselaer Polytechnic Institute as a research fellow. He joined Nanyang Technological University as an Assistant Professor in early 2008. Yan’s research interests mainly include advanced electrode materials for energy storage devices and high-efficiency thermoelectrics. 

 DownLoad:
DownLoad: