| Citation: | Jooheon Sun, Jiseok Kwon, Seunggun Choi, Yeseung Lee, Ungyu Paik, Taeseup Song. In-situ surface vanadate anchoring strategy for robust and efficient seawater oxidation[J]. Materials Lab, 2025, 4(3): 250006. doi: 10.54227/mlab.20250006 |
In-situ surface vanadate anchoring strategy for robust and efficient seawater oxidation
-
Abstract
Direct seawater electrolysis is a promising alternative for carbon-neutral energy systems since large-scale hydrogen production is limited by the cost and scarcity of ultrapure water. Yet the high concentration of Cl− in seawater critically undermines electrocatalyst stability. Herein, we report a novel vanadate-anchoring strategy that simultaneously boosts the activity and durability of Ni(OH)2 anode for alkaline seawater oxidation. Electrochemical anchoring of vanadate onto the Ni(OH)2 surface enables facile surface reconstruction, which significantly modulates the electronic structure of the electrocatalyst and adsorption ability. The tailored electronic structure induced by the anchoring of vanadate steers the reaction along a lattice oxygen-mediated mechanism (LOM). Furthermore, anchored vanadate endows with electrostatic repulsion toward Cl−, granting durability for direct seawater electrolysis. The optimized vanadate-anchored electrocatalyst, Ni(OH)2-Vi, requires only 284 mV at 10 mA cm−2 in simulated alkaline seawater, surpassing 328 mV of Ni(OH)2 and 314 mV of Ni(OH)2-Pi. Also during a 200 h chronopotentiometry at 100 mA cm−2, Ni(OH)2-Vi shows a degradation rate of only 0.198 mV h−1, markedly lower than the 0.812 mV h−1 for Ni(OH)2 and 0.780 mV h−1 for Ni(OH)2-Pi.
-
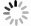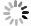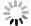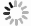
-
References
1. S. Zheng, K. Liu, Y. Li, B. Li, A. Usman, Renew. Energy, 2024, 227, 120465 2. J. A. Turner, Science, 2004, 305, 972 3. S. Evro, B. A. Oni, O. S. Tomomewo, Int. J. Hydrog. Energy, 2024, 78, 1449 4. M. S. Dresselhaus, I. Thomas, Nature, 2001, 414, 332 5. J. Kwon, H. Han, S. Choi, K. Park, S. Jo, U. Paik, T. Song, ChemCatChem, 2019, 11, 5898 6. J. Kwon, S. Sun, S. Choi, K. Lee, S. Jo, K. Park, Y. K. Kim, H. B. Park, H. Y. Park, J. H. Jang, Adv. Mater., 2023, 35, 2300091 7. S. Choi, J. Kwon, C. Park, K. Park, H. B. Park, U. Paik, T. Song, Energy & Fuels, 2023, 37, 18128 8. S. Choi, J. Kwon, S. Jo, S. Kim, K. Park, S. Kim, H. Han, U. Paik, T. Song, Appl. Catal. B, 2021, 298, 120530 9. L. OhIsson, Phys. Chem. Earth, Part B, 2000, 25, 213 10. T. Younos, J. Contemp. Water Res. Educ., 2005, 132, 39 11. M. Khan, T. Al-Attas, S. Roy, M. M. Rahman, N. Ghaffour, V. Thangadurai, S. Larter, J. Hu, P. M. Ajayan, M. G. Kibria, Energy Environ. Sci. , 2021, 14, 4831 12. S. Zhang, Y. Wang, S. Li, Z. Wang, H. Chen, L. Yi, X. Chen, Q. Yang, W. Xu, A. Wang, Nat. Commun., 2023, 14, 4822 13. P. Guo, D. Liu, R. Wu, Small Struct., 2023, 4, 2300192 14. S. Zhang, W. Xu, H. Chen, Q. Yang, H. Liu, S. Bao, Z. Tian, E. Slavcheva, Z. Lu, Adv. Mater., 2024, 36, 2311322 15. L. Yu, Q. Zhu, S. Song, B. McElhenny, D. Wang, C. Wu, Z. Qin, J. Bao, Y. Yu, S. Chen, Nat. commun., 2019, 10, 5106 16. H. You, D. Wu, D. Si, M. Cao, F. Sun, H. Zhang, H. Wang, T. -F. Liu, R. Cao, J. Am. Chem. Soc., 2022, 144, 9254 17. S. r. Dresp, F. Dionigi, M. Klingenhof, P. Strasser, ACS Energy Lett., 2019, 4, 933 18. Z. Li, Y. Yao, S. Sun, J. Liang, S. Hong, H. Zhang, C. Yang, X. Zhang, Z. Cai, J. Li, Angew. Chem. Int. Ed., 2024, 63, e202316522 19. X. Zhang, H. Zhang, Z. Chen, X. Chen, J. Wang, S. Wei, S. Liu, Z. Wang, F. Dai, M. Wang, Adv. Funct. Mater., 2024, 1, 2418940 20. L. Shao, X. Han, L. Shi, T. Wang, Y. Zhang, Z. Jiang, Z. Yin, X. Zheng, J. Li, X. Han, Adv. Energy Mater., 2024, 14, 2303261 21. Z. He, M. Ajmal, M. Zhang, X. Liu, Z. F. Huang, C. Shi, R. Gao, L. Pan, X. Zhang, J. J. Zou, Adv. Sci., 2023, 10, 2304071 22. Y. Tang, C. Wu, Q. Zhang, H. Zhong, A. Zou, J. Li, Y. Ma, H. An, Z. Yu, S. Xi, Angew. Chem. Int. Ed., 2023, 135, e202309107 23. H. Liao, G. Ni, P. Tan, K. Liu, X. Liu, H. Liu, K. Chen, X. Zheng, M. Liu, J. Pan, Adv. Mater., 2023, 35, 2300347 24. J. Kwon, H. Han, S. Jo, S. Choi, K. Y. Chung, G. Ali, K. Park, U. Paik, T. Song, Adv. Energy Mater., 2021, 11, 2100624 25. T. Li, X. Zhao, M. G. Sendeku, X. Zhang, L. Xu, Z. Wang, S. Wang, X. Duan, H. Liu, W. Liu, Chem. Eng. J., 2023, 460, 141413 26. A. K. Singh, S. Mukherjee, J. P. Singh, A. Gupta, P. Singh, J. Power Sources, 2025, 658, 238330 27. K. H. Shin, J. Park, S. K. Park, P. Nakhanivej, S. M. Hwang, Y. Kim, H. S. Park, J. Ind. Eng. Chem., 2019, 72, 250 28. W. Shao, M. Xiao, C. Yang, M. Cheng, S. Cao, C. He, M. Zhou, T. Ma, C. Cheng, S. Li, Small, 2022, 18, 2105763 29. R. Kumar, T. Bhuvana, P. Rai, A. Sharma, J. Electrochem. Soc., 2018, 165, B1 30. W. Yang, F. Qi, W. An, H. Yu, S. Liu, P. Ma, R. Chen, S. Liu, L. -L. Lou, K. Yu, ACS Catal., 2024, 14, 5936 31. H. H. Kristoffersen, T. Vegge, H. A. Hansen, Chem. Sci., 2018, 9, 6912 32. J. Chen, H. Chen, T. Yu, R. Li, Y. Wang, Z. Shao, S. Song, Electrochem. Energy Rev., 2021, 4, 566 33. L. Gao, X. Cui, C. D. Sewell, J. Li, Z. Lin, Chem. Soc. Rev., 2021, 50, 8428 34. Y. Zeng, M. Zhao, Z. Huang, W. Zhu, J. Zheng, Q. Jiang, Z. Wang, H. Liang, Adv. Energy Mater., 2022, 12, 2201713 35. Z. Xu, M. Zhong, P. Liu, Z. Zhang, J. Energy Chem., 2025, 106, 123 36. Y. Li, Y. Wu, M. Yuan, H. Hao, Z. Lv, L. Xu, B. Wei, Appl. Catal. B, 2022, 318, 121825 37. X. Wang, H. Zhong, S. Xi, W. S. V. Lee, J. Xue, Adv. Mater., 2022, 34, 2107956 38. N. Zhang, Y. Chai, Energy Environ. Sci., 2021, 14, 4647 39. H. Zhong, Q. Zhang, J. Yu, X. Zhang, C. Wu, H. An, Y. Ma, H. Wang, J. Zhang, Y. -W. Zhang, Nat. Commun., 2023, 14, 7488 40. X. Gao, Y. Zhu, S. Yang, W. Zhang, R. Cao, J. Energy Chem., 2025, 103, 393 41. J. Cai, D. Qu, X. He, P. Su, Q. Dong, Y. Li, B. Zhu, S. Yu, Electrochim. Acta, 2024, 507, 145067 42. C. Yang, Z. Cai, J. Liang, K. Dong, Z. Li, H. Sun, S. Sun, D. Zheng, H. Zhang, Y. Luo, Nano Res., 2024, 17, 5786 43. M. Yu, J. Li, F. Liu, J. Liu, W. Xu, H. Hu, X. Chen, W. Wang, F. Cheng, J. Energy Chem., 2022, 72, 361 44. H. Wang, N. Jiang, B. Huang, Q. Yu, L. Guan, EES Catal., 2024, 2, 1092 45. C. Yang, C. Laberty-Robert, D. Batuk, G. Cibin, A. V. Chadwick, V. Pimenta, W. Yin, L. Zhang, J. -M. Tarascon, A. Grimaud, J. Phys. Chem. Lett., 2017, 8, 3466 46. X. Kang, F. Yang, Z. Zhang, H. Liu, S. Ge, S. Hu, S. Li, Y. Luo, Q. Yu, Z. Liu, Nat. Commun., 2023, 14, 3607 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
-
Supplementary Information
Supplementary+Information 
Information
Article Metrics
-
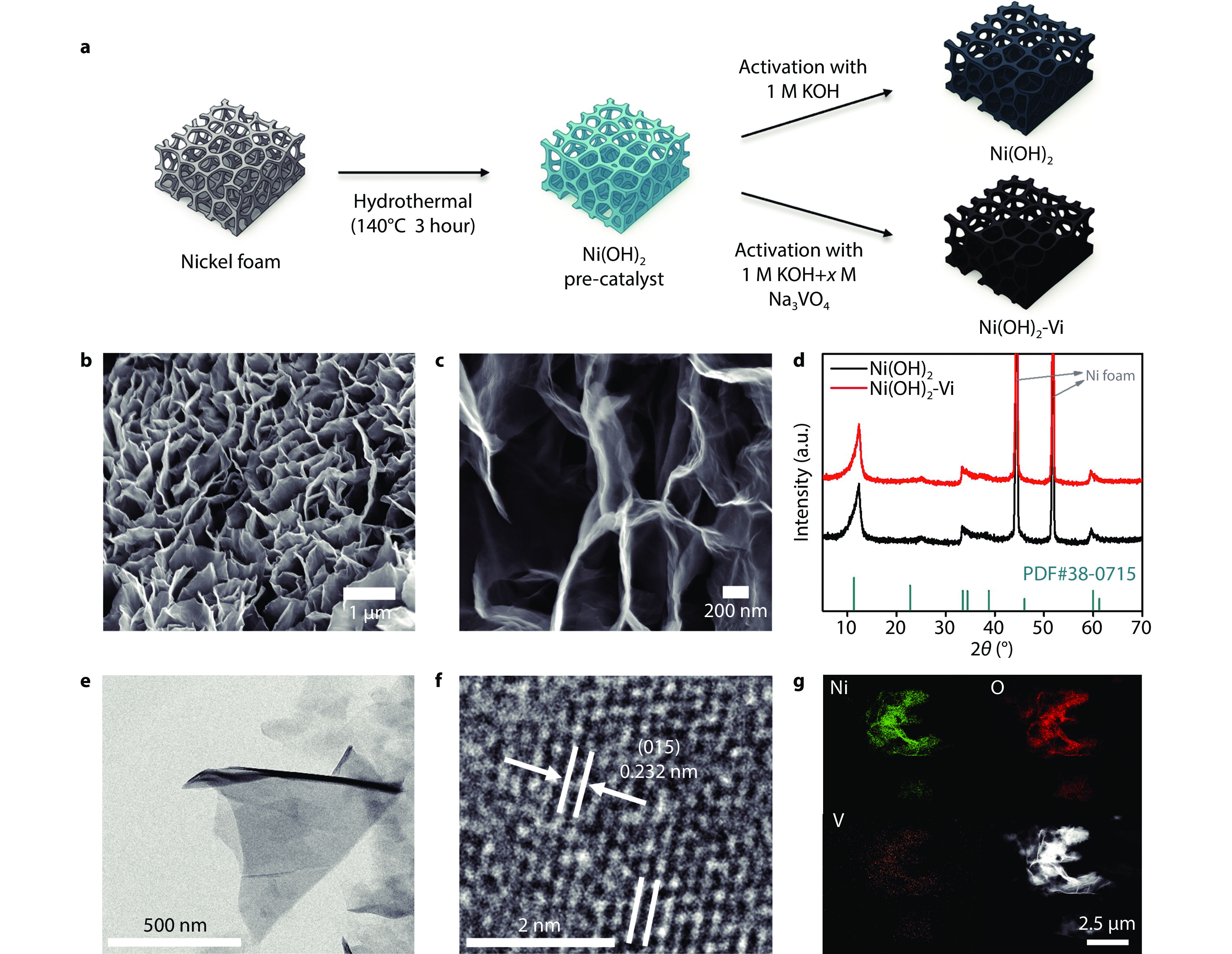
Figure 1.
Preparation and material characterizations of the Ni(OH)2-Vi. a Schematic illustration of the preparation for Ni(OH)2-Vi. b, c Scanning electron microscopy (SEM) images, d X-ray diffraction (XRD) patterns of Ni(OH)2 and Ni(OH)2-Vi. e Transmission electron microscopy (TEM) image of Ni(OH)2-Vi. f High-resolution transmission electron microscopy (HR-TEM) image of Ni(OH)2-Vi. g Energy-dispersive X-ray spectroscopy (EDS) elemental mapping of Ni(OH)2-Vi.
-
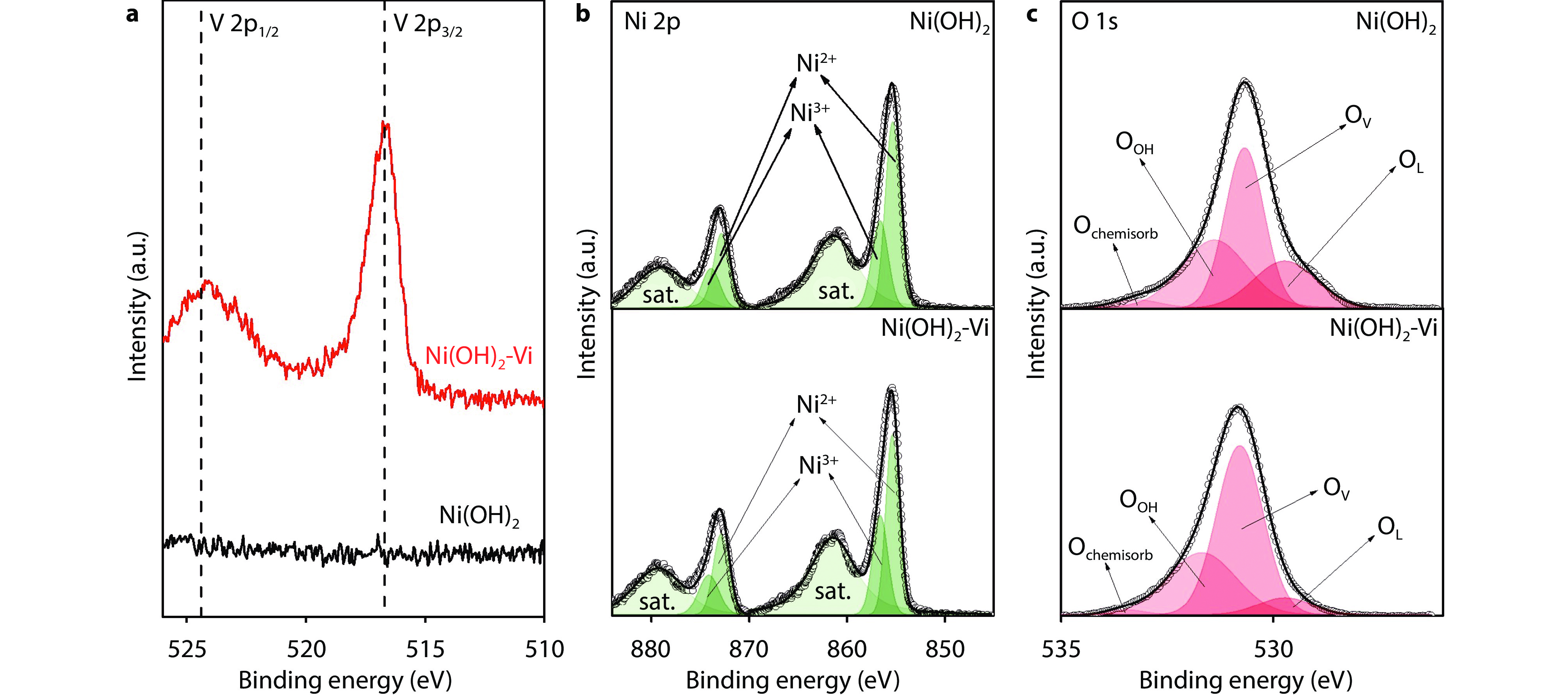
Figure 2.
X-ray photoelectron spectroscopy (XPS) spectra of the Ni(OH)2 and Ni(OH)2-Vi. a V 2p spectra of Ni(OH)2 and Ni(OH)2-Vi. b Ni 2p spectra of Ni(OH)2 and Ni(OH)2-Vi. c O 1s spectra of Ni(OH)2 and Ni(OH)2-Vi.
-
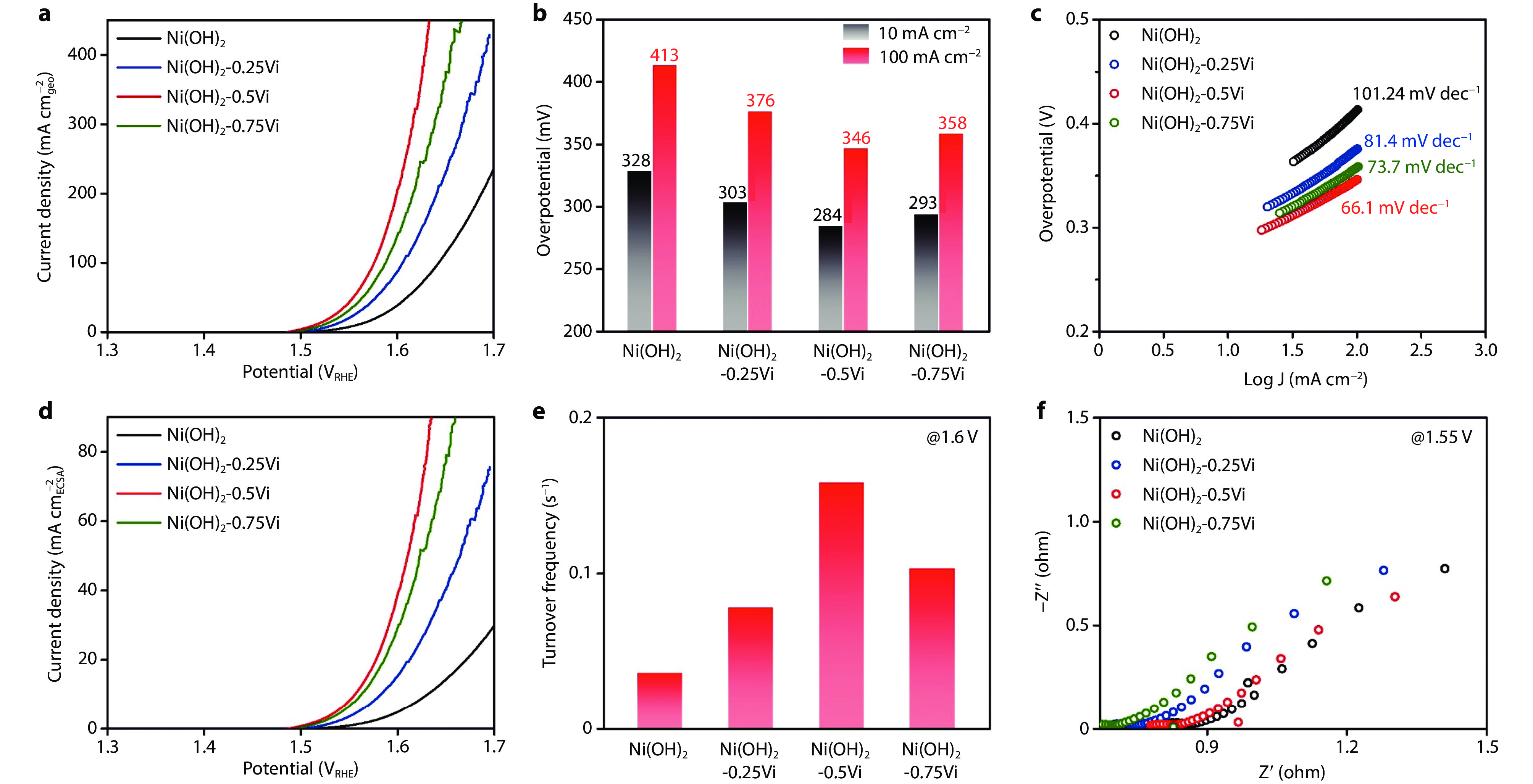
Figure 3.
Electrochemical seawater oxidation property of the Ni(OH)2-xVi. a Polarization curves, b Corresponding overpotentials at 10 and 100 mA cm−2, c Tafel plots. d Electrochemical surface area (ECSA) normalized polarization curves. e Turnover frequency of Ni(OH)2-xVi. f Nyquist plot at 1.55VRHE of Ni(OH)2-xVi.
-
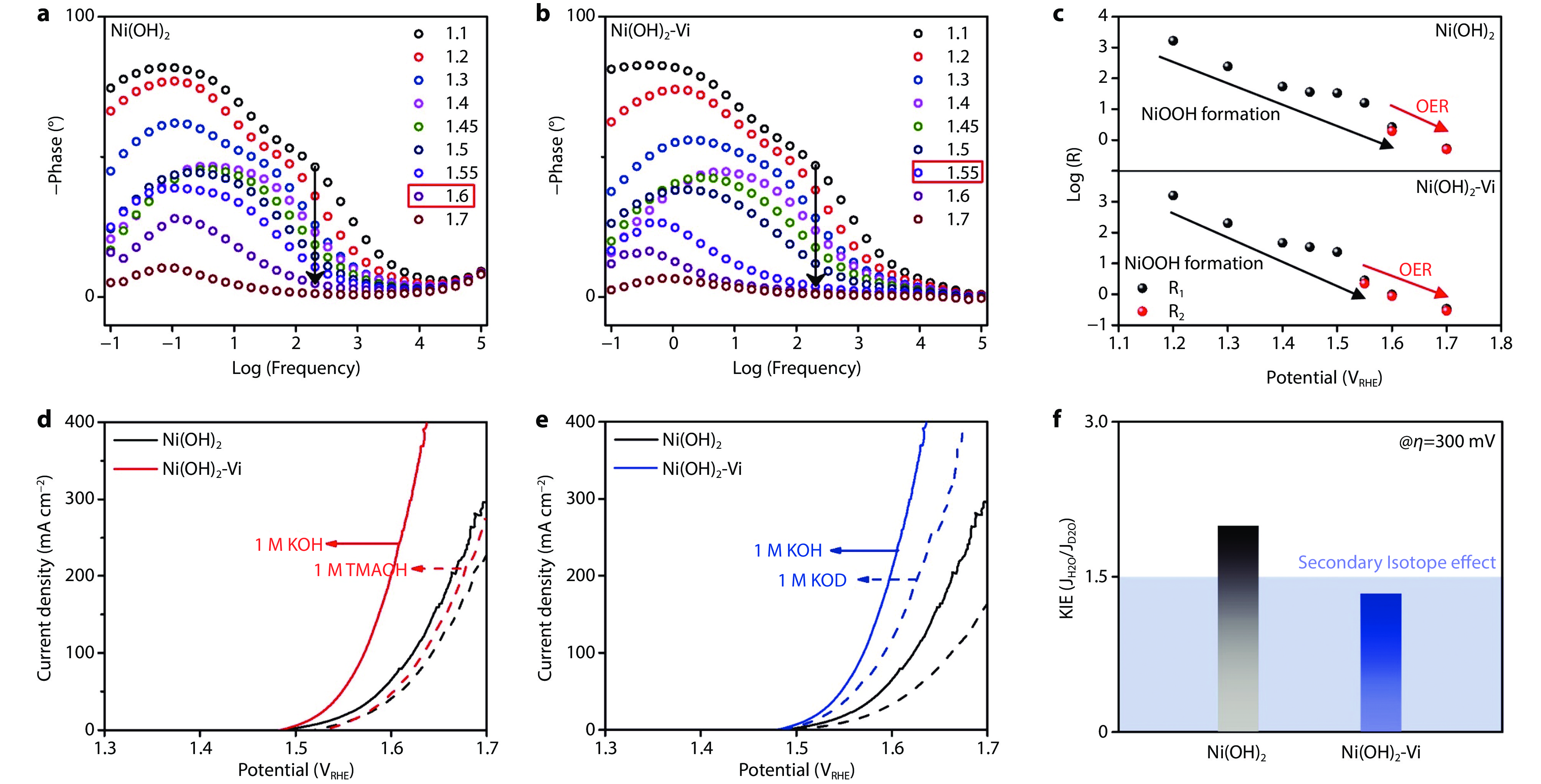
Figure 4.
Mechanism study of vanadate anchoring-triggered OER activity enhancement. a, b Operando bode plots during activation process of Ni(OH)2 and Ni(OH)2-Vi. c Correlation of the equivalent resistance and potentials fitted by Nyquist plot during activation process of Ni(OH)2 and Ni(OH)2-Vi. d Polarization curves measured with 1M KOH and 1M TMAOH of Ni(OH)2 and Ni(OH)2-Vi. e Polarization curves measured with 1M KOH and 1M KOD f The kinetic isotope effect of Ni(OH)2 and Ni(OH)2-Vi.
-
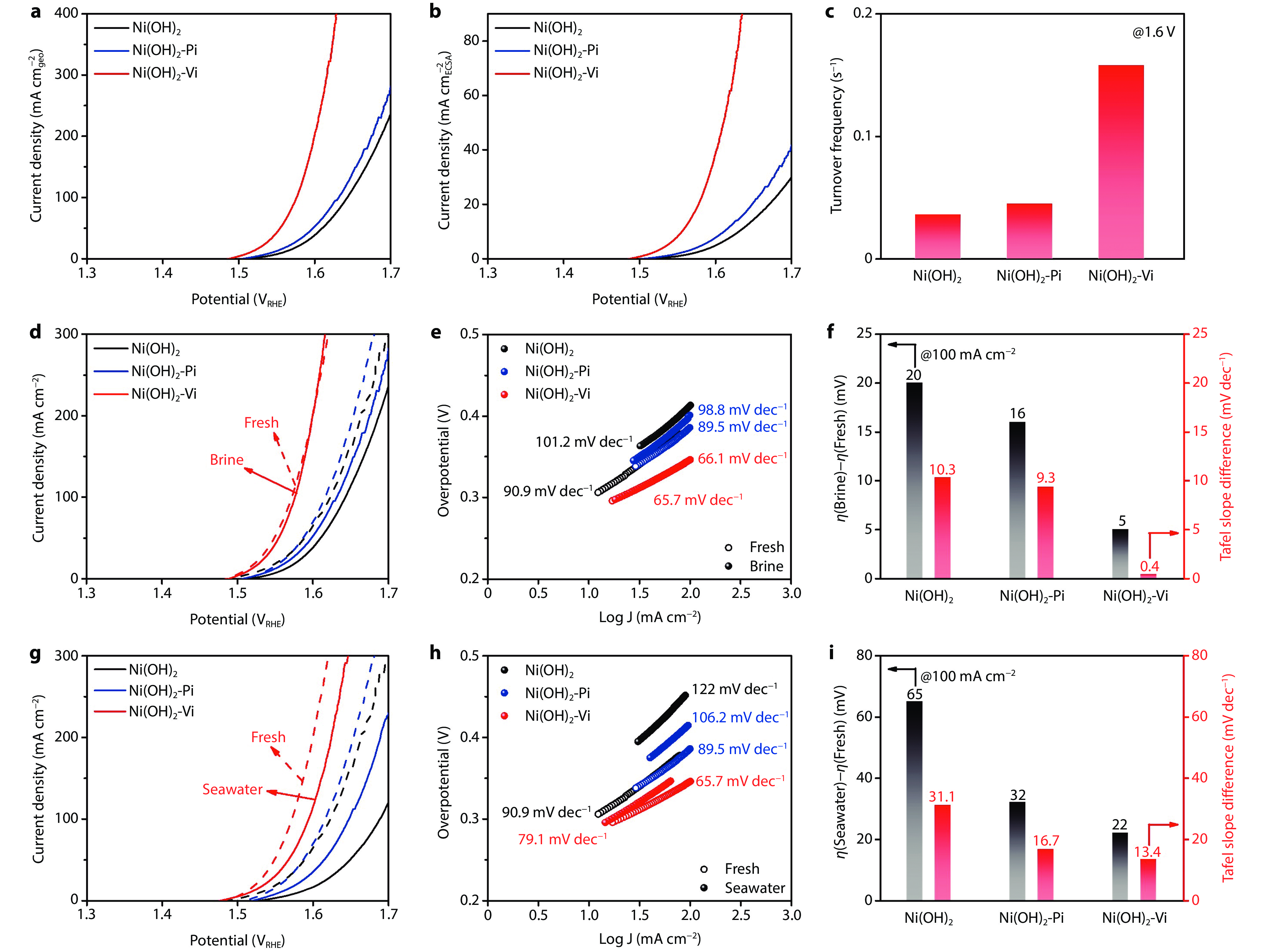
Figure 5.
Superior seawater oxidation property of vanadate anchoring strategy over phosphate anchoring strategy a Polarization curves comparing Ni(OH)2-Vi and Ni(OH)2-Pi. b Electrochemical surface area (ECSA) normalized polarization curves comparing Ni(OH)2-Vi and Ni(OH)2-Pi. c Turnover frequency comparing Ni(OH)2-Vi and Ni(OH)2-Pi. d Polarization curves measured with fresh electrolyte (1 M KOH) and brine electrolyte (1 M KOH + 0.5 M NaCl) and e corresponding Tafel plots. f Activity variation of overpotential and Tafel slopes under fresh electrolyte and brine electrolyte. g Polarization curves measured with fresh electrolyte and alkaline seawater (1 M KOH + seawater) and h corresponding Tafel plots. i Activity variation of overpotential and Tafel slopes under fresh electrolyte and alkaline seawater.
-
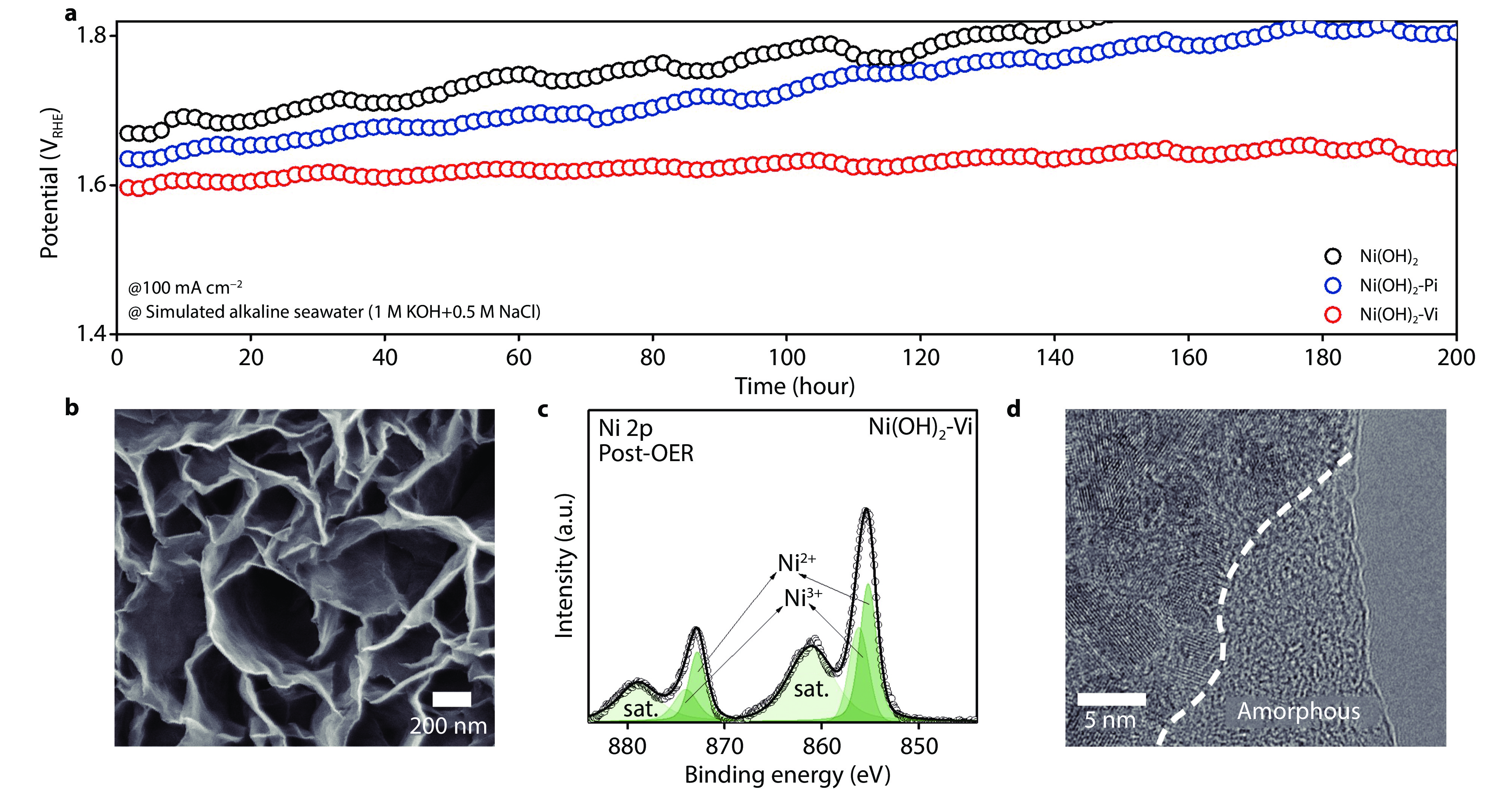
Figure 6.
Electrochemical stability in alkaline seawater and ex-situ characterization a Chronopotentiometry OER stability test at current density of 100 mA/cm2 for 200 h under simulated alkaline seawater. b Post-OER SEM image of Ni(OH)2-Vi. c Post-OER Ni 2p XPS spectra of Ni(OH)2-Vi. d Post-OER HR-TEM image of Ni(OH)2-Vi.


 DownLoad:
DownLoad: