| Citation: | Pengyuan Zhang, Ming Liu, Yu-Ke Zhu, Jiehe Sui. A method for the direct measurement of chemical bonds[J]. Materials Lab, 2025, 4(3): 250009. doi: 10.54227/mlab.20250009 |
A method for the direct measurement of chemical bonds
-
Abstract
-
-
References
1. Y. Yu, M. Cagnoni, O. Cojocaru-Mirédin, M. Wuttig, Adv. Funct. Mater., 2020, 30, 1904862 2. O. Cojocaru-Mirédin, Y. Yu, J. Köttgen, T. Ghosh, C. F. Schön, S. Han, C. Zhou, M. Zhu, M. Wuttig, Adv. Mater., 2024, 36, 2403046 3. M. Zhu, O. Cojocaru-Mirédin, A. M. Mio, J. Keutgen, M. Küpers, Y. Yu, J. Y. Cho, R. Dronskowski, M. Wuttig, Adv. Mater., 2018, 30, 1706735 4. Y. Yu, O. Cojocaru-Mirédin, M. Wuttig, Phys. Status Solidi A, 2023, 221, 2300425 5. M. Wuttig, V. L. Deringer, X. Gonze, C. Bichara, J. Y. Raty, Adv. Mater., 2018, 30, 1803777 6. M. Liu, M. Guo, H. Lyu, Y. Lai, Y. Zhu, F. Guo, Y. Yang, K. Yu, X. Dong, Z. Liu, W. Cai, M. Wuttig, Y. Yu, J. Sui, Nat. Commun., 2024, 15, 8286 7. W. J. Yang, T. Ha, B. C. Park, K. -S. Jeong, J. Y. Park, D. Kim, C. Lee, J. Park, M. -H. Cho, ACS Nano, 2022, 16, 2024 8. W. Zhang, H. Zhang, S. Sun, X. Wang, Z. Lu, X. Wang, J. J. Wang, C. Jia, C. F. Schön, R. Mazzarello, Adv. Sci., 2023, 10, 2300901 9. Y. Yu, C. Zhou, T. Ghosh, C. F. Schön, Y. Zhou, S. Wahl, M. Raghuwanshi, P. Kerres, C. Bellin, A. Shukla, Adv. Mater., 2023, 35, 2300893 10. H. Luo, X. -L. Shi, Y. Liu, M. Li, M. Zhang, X. Luo, M. Wang, X. Huang, L. Hu, Z. -G. Chen, Nat. Commun., 2025, 16, 3136 11. Y. Liu, X. Zhang, P. Nan, B. Zou, Q. Zhang, Y. Hou, S. Li, Y. Gong, Q. Liu, B. Ge, O. Cojocaru-Mirédin, Y. Yu, Y. Zhang, G. Chen, M. Wuttig, G. Tang, Adv. Funct. Mater., 2022, 32, 2209980 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
Figure 1.
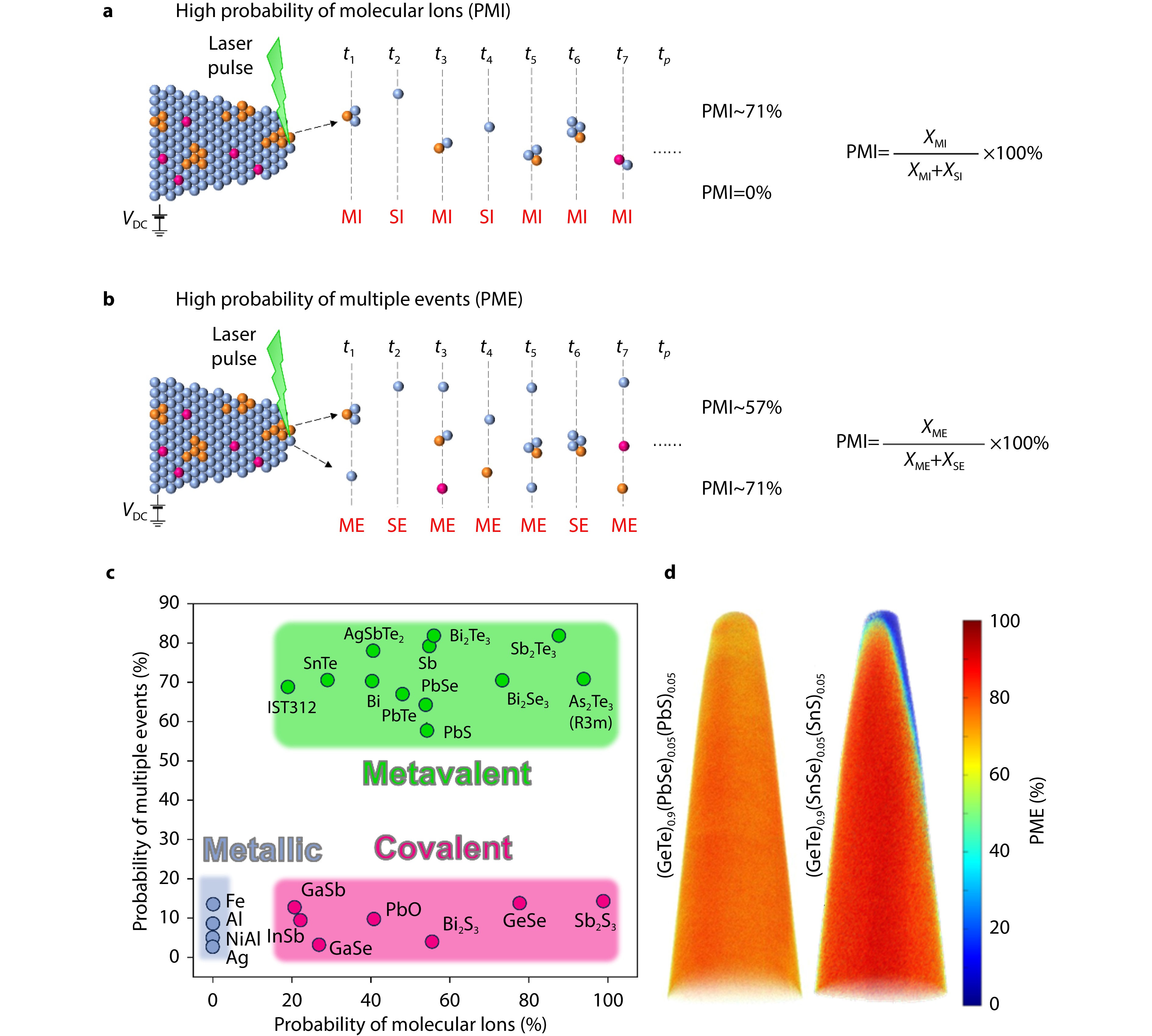
Schematic diagram of APT localized chemical bond characterization. a Sketch of a bond-breaking experiment for a sequence of successful pulses exhibiting high PMI, as well as b a sketch of the same experiment exhibiting high PME. SI and MI respectively stand for a single ion and molecular ions, while SE and ME respectively stand for a single event and multiple events to be dislodged upon bond rupture. c Demarcation of different chemical bonding mechanisms by PMI and PME: metals (in blue), metavalent solids (in green), and covalently bonded compounds (in red). d The corresponding bonding mechanisms of (GeTe)0.9(PbSe)0.05(PbS)0.05 and (GeTe)0.9(SnSe)0.05(SnS)0.05 are revealed by 3D PME maps characterized by APT.


 DownLoad:
DownLoad:

