| Citation: | Yu Wu, Matthew F. Webster, Brian Joy, Steve Beyer, David Kunar, et al. Combined Purification and Crystal Growth of CsPbBr3 by Modified Zone Refining. Materials Lab 2022, 1, 220019. doi: 10.54227/mlab.20220019 |
Combined Purification and Crystal Growth of CsPbBr3 by Modified Zone Refining
Published as part of the Virtual Special Issue "Mercouri G. Kanatzidis at 65"
-
Abstract
The all-inorganic semiconducting perovskite Cesium Lead Bromide, CsPbBr3, exhibits promising properties for ionizing radiation detection applications. In this work, polycrystalline CsPbBr3 was synthesized from the melt of binary compounds CsBr and PbBr2. Moisture and oxides in the synthesized CsPbBr3 compounds were removed by a reduction process under hydrogen. The CsPbBr3 materials were purified and grown into high-quality single crystals
via a modified zone refining process. The single-crystal samples obtained from the combined zone-refining/crystal growth process exhibited total trace impurity levels below 1 ppm (w.t. ). Obtained single crystals exhibited an electrical resistivity within a range of 108~109 Ω·cm. Stoichiometry imbalance was observed in the CsPbBr3 crystal growth. Around 1% Cs deficiency was observed in all the samples, despite different ratios between the starting materials of PbBr2 and CsBr. The positive impact of excess PbBr2 in starting materials was also revealed. With a slight excess Pb (2%), CsPbBr3 single crystals displayed significantly higher photosensitivity compared to the stoichiometric or excess Cs samples.-
Keywords:
- Opto-electronic Material /
- Radiation detector /
- Perovskites /
- Compound Semiconductor
-
-
References
1. T. E. Schlesinger, J. E. Toney, H. Yoon, E. Y. Lee, B. A. Brunett, L. Franks, R. B. James, Materials Science and Engineering: R: Reports, 2001, 32, 103 2. S. D. Sordo, L. Abbene, E. Caroli, A. M. Mancini, A. Zappettini, P. Ubertini, Sensors, 2009, 9, 3491 3. H. H. Barrett, J. D. Eskin, H. B. Barber, Physical Review Letters, 1995, 75, 156 4. P. M. Johns, J. C. Nino, Journal of Applied Physics, 2019, 126, 040902 5. A. Mirzaei, J.-S. Huh, S. S. Kim, H. W. Kim, Electronic Materials Letters, 2018, 14, 261 6. L. Gao, Q. Yan, Solar RRL, 2020, 4, 1900210 7. B. D. Milbrath, A. J. Peurrung, M. Bliss, W. J. Weber, Journal of Materials Research, 2008, 23, 2561 8. K. Hitomi, T. Tada, S. Kim, Y. Wu, T. Tanaka, T. Shoji, H. Yamazaki, K. Ishii, IEEE Trans. Nucl. Sci., 2011, 58, 1987 9. C. Szeles, IEEE Trans. Nucl. Sci., 2004, 51, 1242 10. W. Lin, Z. Liu, C. C. Stoumpos, S. Das, Y. He, I. Hadar, J. A. Peters, K. M. McCall, Y. Xu, D. Y. Chung, B. W. Wessels, M. G. Kanatzidis, Crystal Growth & Design, 2019, 19, 4738 11. U. N. Roy, G. S. Camarda, Y. Cui, R. Gul, A. Hossain, G. Yang, J. Zazvorka, V. Dedic, J. Franc, R. B. James, Scientific Reports, 2019, 9, 1620 12. K. Hitomi, T. Shoji, K. Ishii, Journal of Crystal Growth, 2013, 379, 93 13. G. F. Knoll, Radiation Detection and Measurement, John Wiley & Sons, America, 2010. 14. M. Ahmadi, T. Wu, B. Hu, Advanced Materials, 2017, 29, 1605242 15. X. Li, Y. C. Wang, L. Zhu, W. Zhang, H. Q. Wang, J. Fang, ACS Appl. Mater. Interfaces, 2017, 9, 31357 16. Z. Chen, Q. Dong, Y. Liu, C. Bao, Y. Fang, Y. Lin, S. Tang, Q. Wang, X. Xiao, Y. Bai, Y. Deng, J. Huang, Nat. Commun., 2017, 8, 1890 17. B. Han, B. Cai, Q. Shan, J. Song, J. Li, F. Zhang, J. Chen, T. Fang, Q. Ji, X. Xu, H. Zeng, Advanced Functional Materials, 2018, 28, 1804285 18. H. Wei, J. Huang, Nature Communications, 2019, 10, 1066 19. S. Yakunin, D. N. Dirin, Y. Shynkarenko, V. Morad, I. Cherniukh, O. Nazarenko, D. Kreil, T. Nauser, M. V. Kovalenko, Nature Photonics, 2016, 10, 585 20. J. Yu, G. Liu, C. Chen, Y. Li, M. Xu, T. Wang, G. Zhao, L. Zhang, Journal of Materials Chemistry C, 2020, 8, 6326 21. B. Conings, J. Drijkoningen, N. Gauquelin, A. Babayigit, J. D'Haen, L. D'Olieslaeger, A. Ethirajan, J. Verbeeck, J. Manca, E. Mosconi, F. D. Angelis, H.-G. Boyen, Advanced Energy Materials, 2015, 5, 1500477 22. G. P. Nagabhushana, R. Shivaramaiah, A. Navrotsky, Proceedings of the National Academy of Sciences, 2016, 113, 7717 23. C. C. Stoumpos, C. D. Malliakas, J. A. Peters, Z. Liu, M. Sebastian, J. Im, T. C. Chasapis, A. C. Wibowo, D. Y. Chung, A. J. Freeman, B. W. Wessels, M. G. Kanatzidis, Crystal Growth & Design, 2013, 13, 2722 24. Y. He, L. Matei, H. J. Jung, K. M. McCall, M. Chen, C. C. Stoumpos, Z. Liu, J. A. Peters, D. Y. Chung, B. W. Wessels, M. R. Wasielewski, V. P. Dravid, A. Burger, M. G. Kanatzidis, Nature Communications, 2018, 9, 1609 25. J. Kang, L.-W. Wang, The Journal of Physical Chemistry Letters, 2017, 8, 489 26. J. Ding, S. Du, Z. Zuo, Y. Zhao, H. Cui, X. Zhan, The Journal of Physical Chemistry C, 2017, 121, 4917 27. B. Murali, H. K. Kolli, J. Yin, R. Ketavath, O. M. Bakr, O. F. Mohammed, ACS Materials Letters, 2020, 2, 184 28. X. Cheng, S. Yang, B. Cao, X. Tao, Z. Chen, Advanced Functional Materials, 2020, 30, 1905021 29. D. Y. Chung, M. G. Kanatzidis, F. Meng, C. D. Malliakas, SPIE Optics + Photonics, San Diego, CA, USA, August 2016. 30. M. Zhang, Z. Zheng, Q. Fu, Z. Chen, J. He, S. Zhang, C. Chen, W. Luo, Journal of Crystal Growth, 2018, 484, 37 31. K. Wang, L. Jing, Q. Yao, J. Zhang, X. Cheng, Y. Yuan, C. Shang, J. Ding, T. Zhou, H. Sun, W. Zhang, H. Li, The Journal of Physical Chemistry Letters, 2021, 12, 1904 32. J.-L. Pouchou, F. Pichoir, Electron Probe Quantitation, Springer, Germany, 1991. 33. K. F. J. Heinrich, 11th Int. Congr. on X-Ray Optics and Microanalysis, London, Ontario, Canada, August 1986. 34. K. L. Williams, An introduction to X-ray spectrometry: X-ray fluorescence and electron microprobe analysis, Allen & Unwin Boston, America, 1987. 35. H. Zhang, F. Wang, Y. Lu, Q. Sun, Y. Xu, B.-B. Zhang, W. Jie, M. G. Kanatzidis, Journal of Materials Chemistry C, 2020, 8, 1248 36. H. Kim, Y. Ogorodnik, A. Kargar, L. Cirignano, C. L. Thrall, W. Koehler, S. P. O'Neal, Z. He, E. Swanberg, S. A. Payne, M. R. Squillante, K. Shah, Frontiers in Physics, 2020, 8, 55 37. A. Churilov, W. Higgins, G. Ciampi, H. Kim, L. Cirignano, F. Olschner, K. Shah, SPIE Optics+Photonics 2008, San Diego, CA, USA, August 2008. 38. U. Makanda, A. Voinot, R. Kandel, Y. Wu, M. Leybourne, P. Wang, Journal of Analytical Atomic Spectrometry, 2020, 35, 2672 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
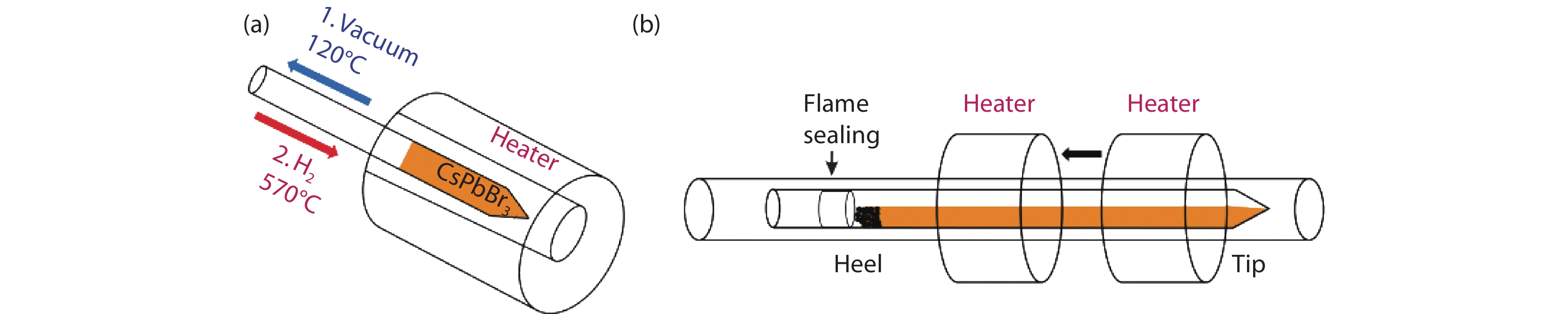
Figure 1.
System design for (a) H2 reduction and (b) Horizontal zone refining furnace.
-
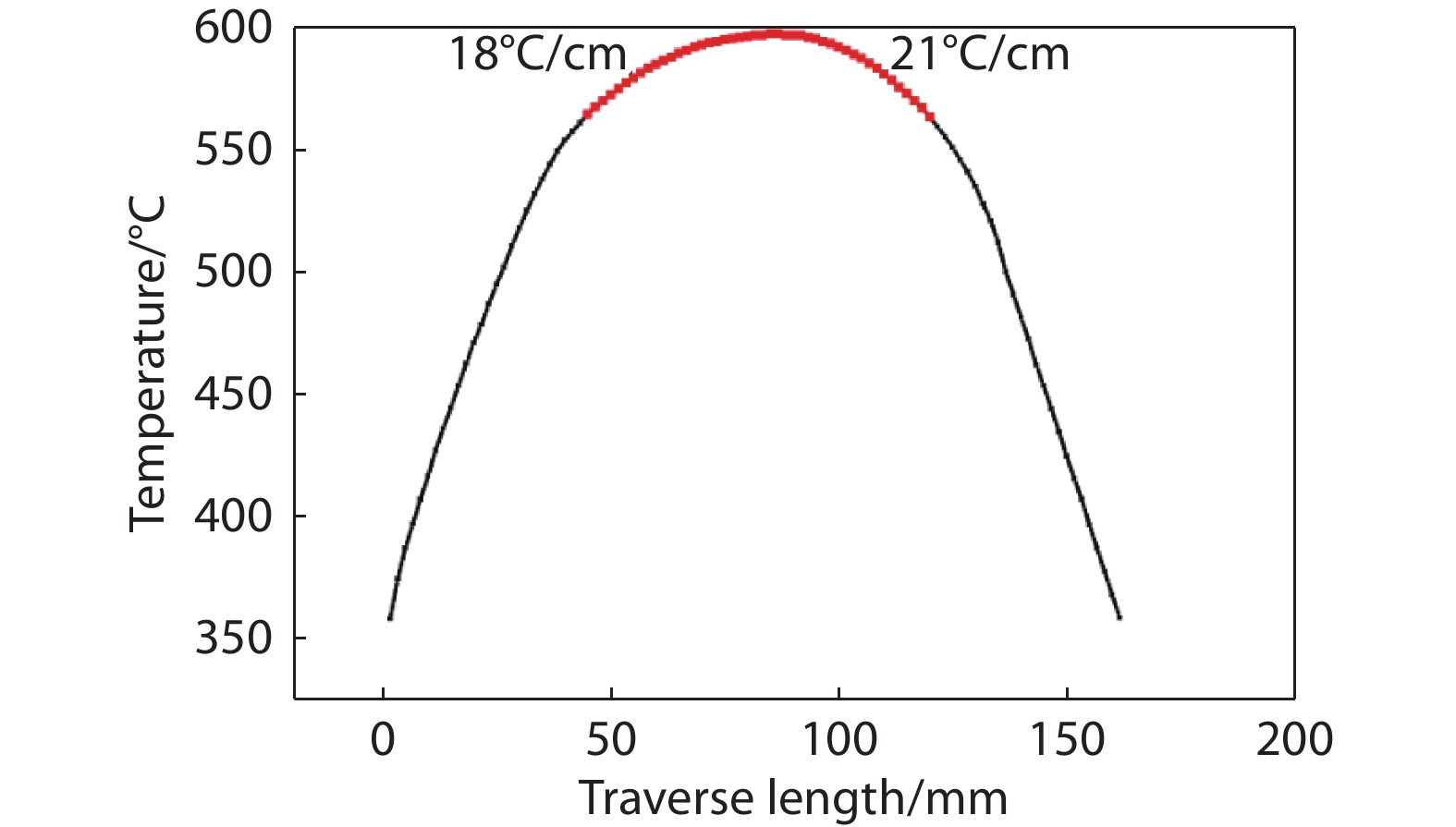
Figure 2.
Temperature profile for a typical zone-refining cycle, where the red region marks the temperature above the melting point (567
$ {}^\circ \text{C}$ -
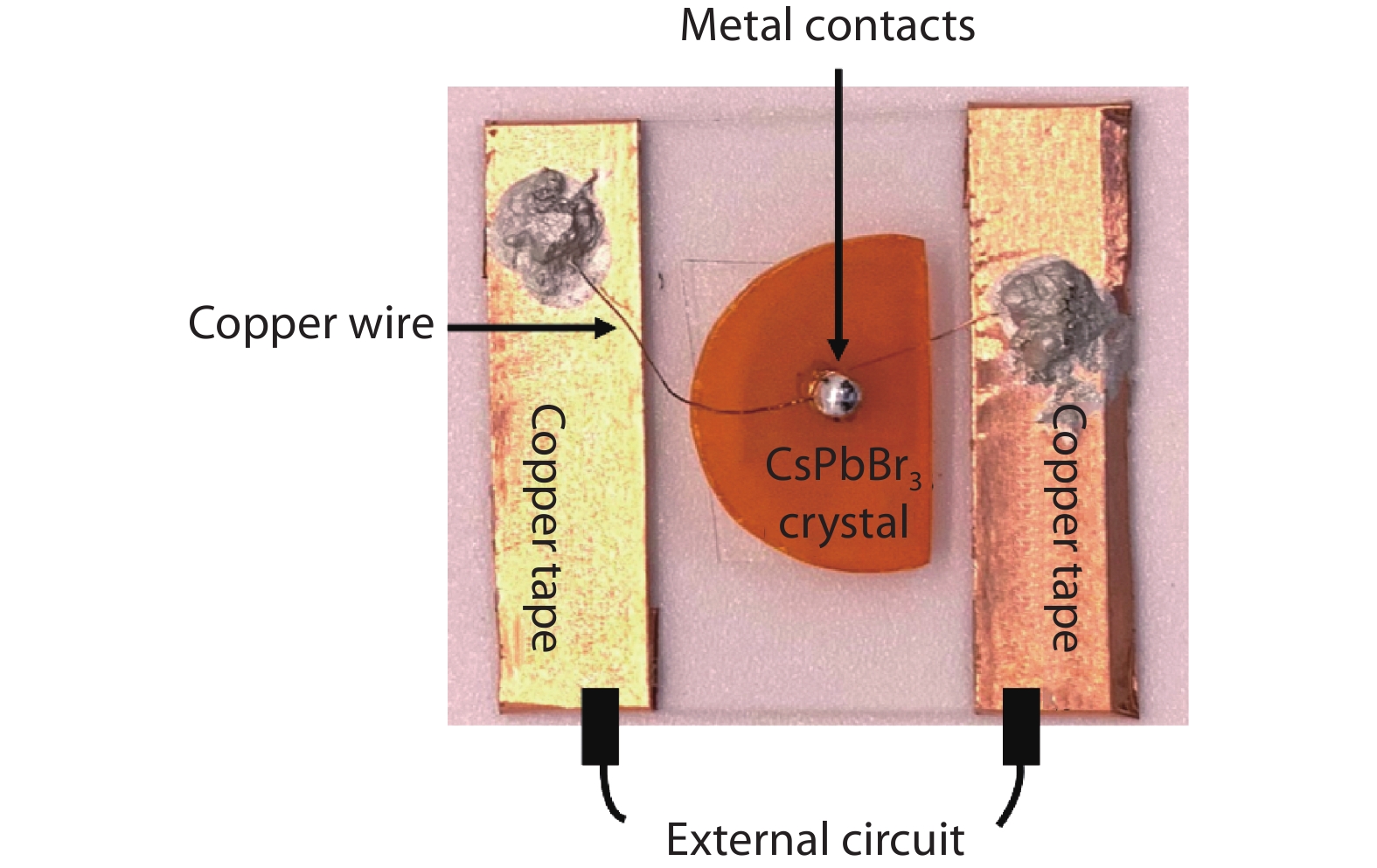
Figure 3.
Main components of a fabricated CsPbBr3 test device.
-
Figure 4.
Comparison of zone refining performances between two sealing techniques (a) sealing under a high vacuum, (b) sealing with 0.3 atm of H2 gas.
-
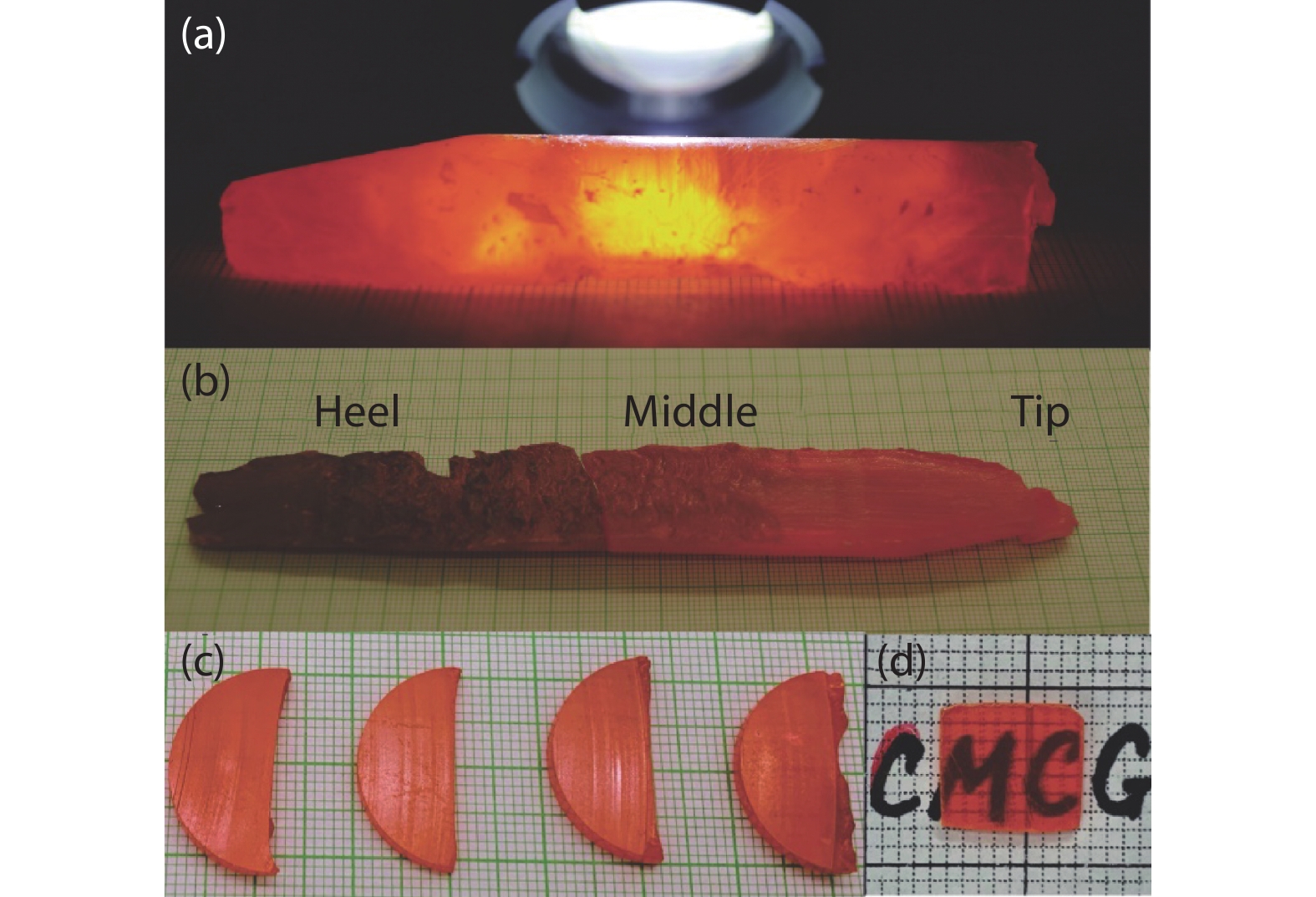
Figure 5.
(a) and (b) CsPbBr3 bulk after zone refining. (c) As-cut crystal wafers. (d) The polished detector.
-
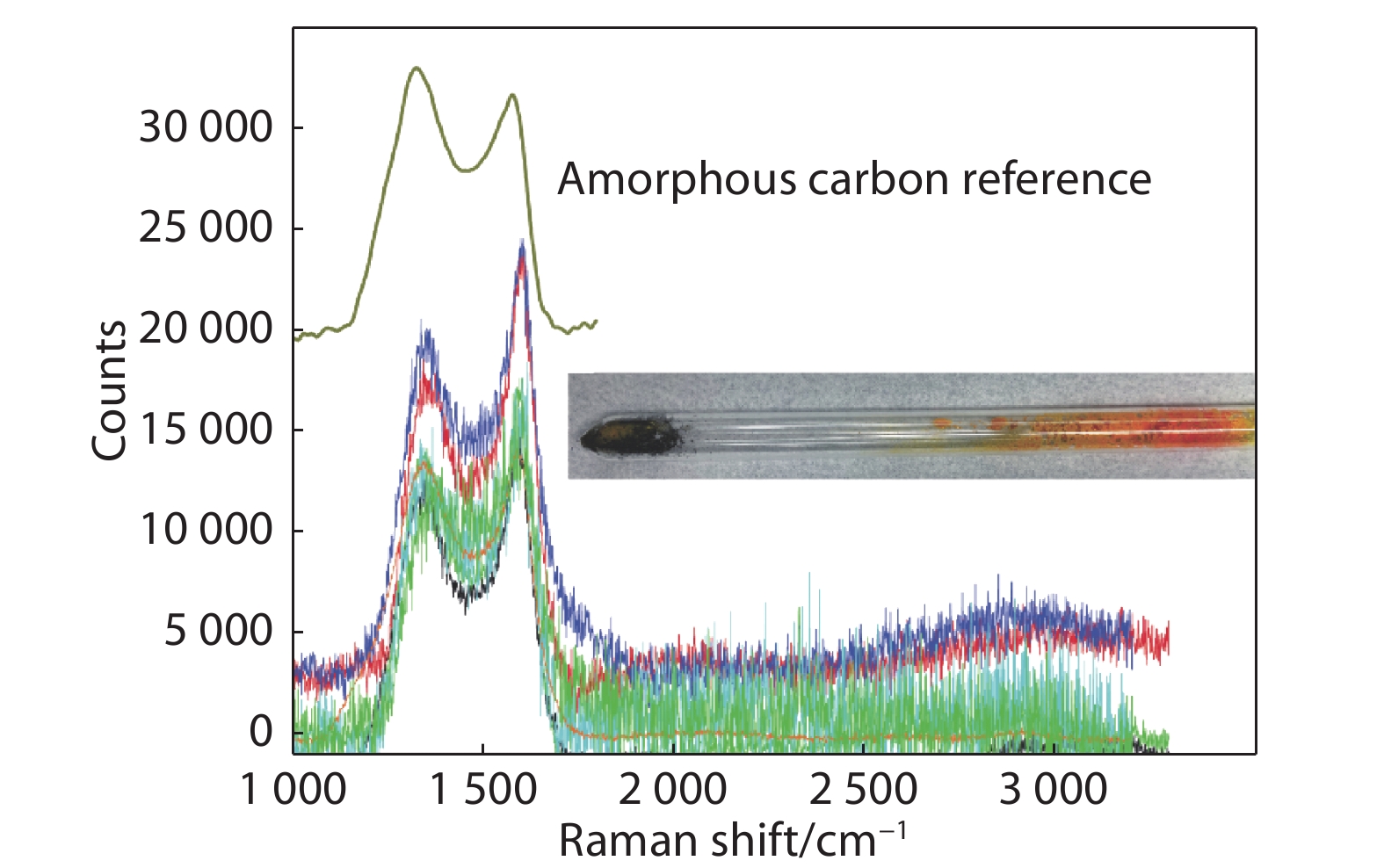
Figure 6.
Raman spectra of black impurity. Insert shows the isolation of the black impurity from CsPbBr3 by sublimation.
-
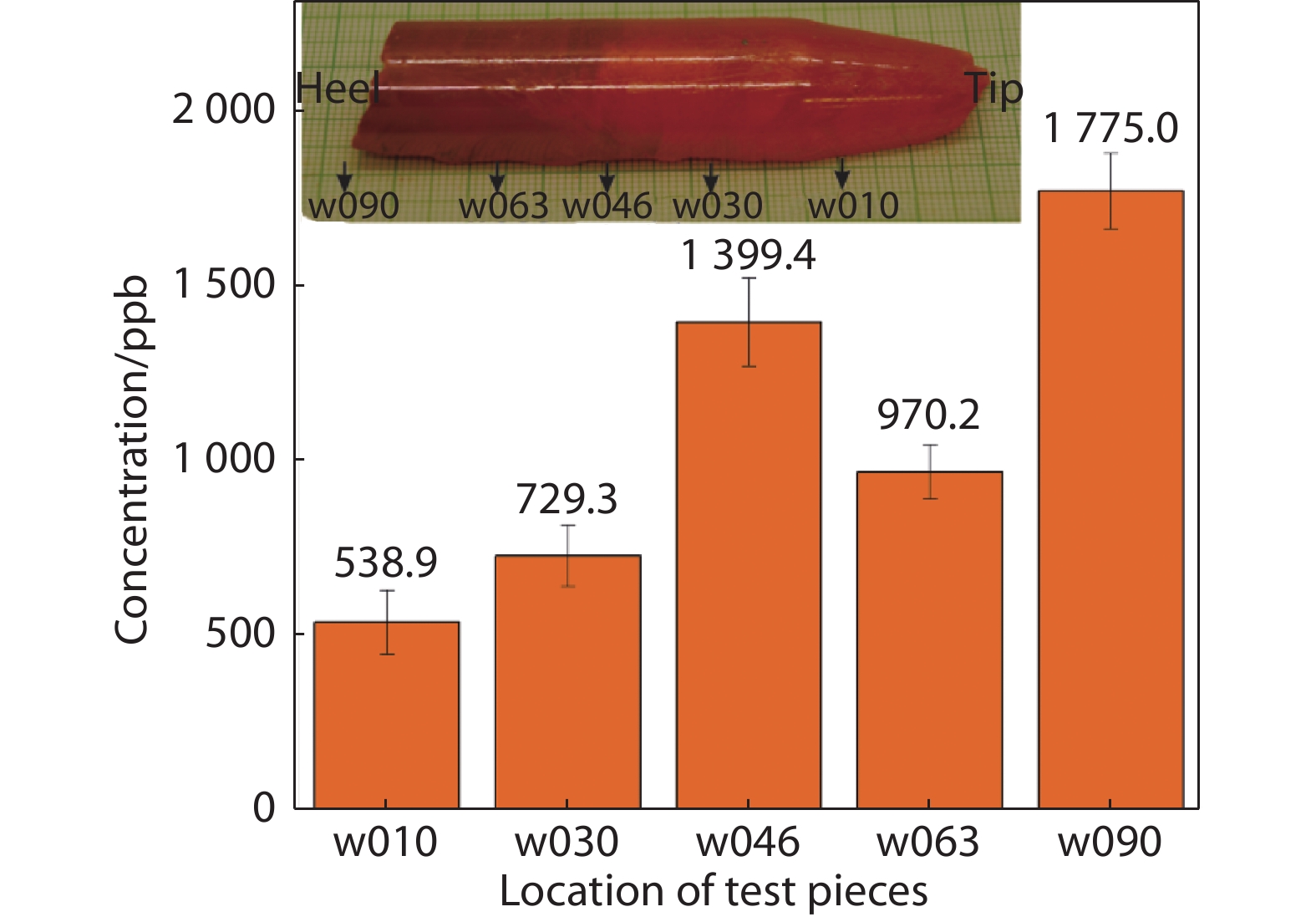
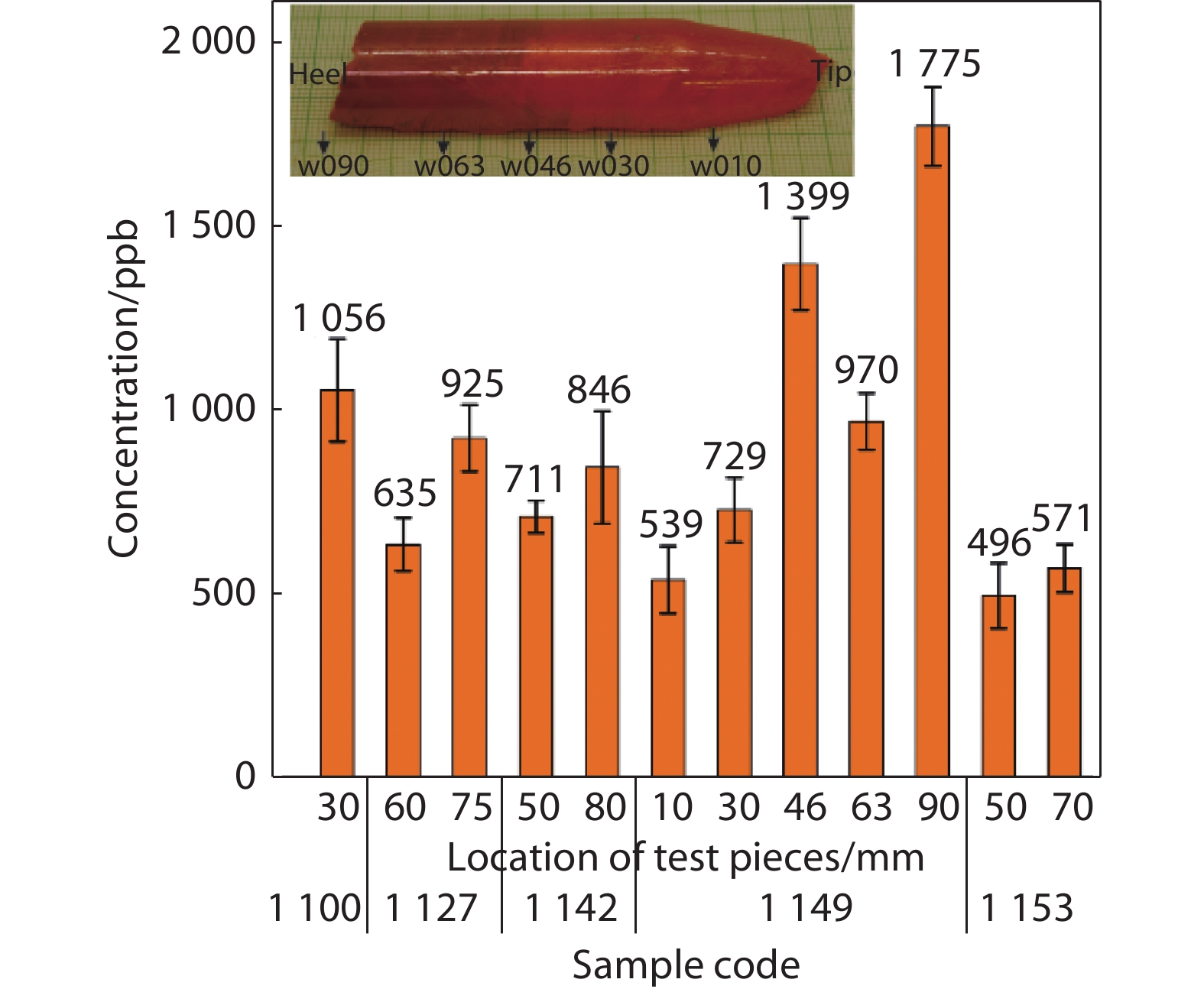
Figure 7.
Total impurity levels of samples
1149 at different positions along with the crystal bulk. Insert shows the position of testing pieces taken from sample1149 , e.g., ‘w010’ indicates 10 mm from the tip of the bulk. -
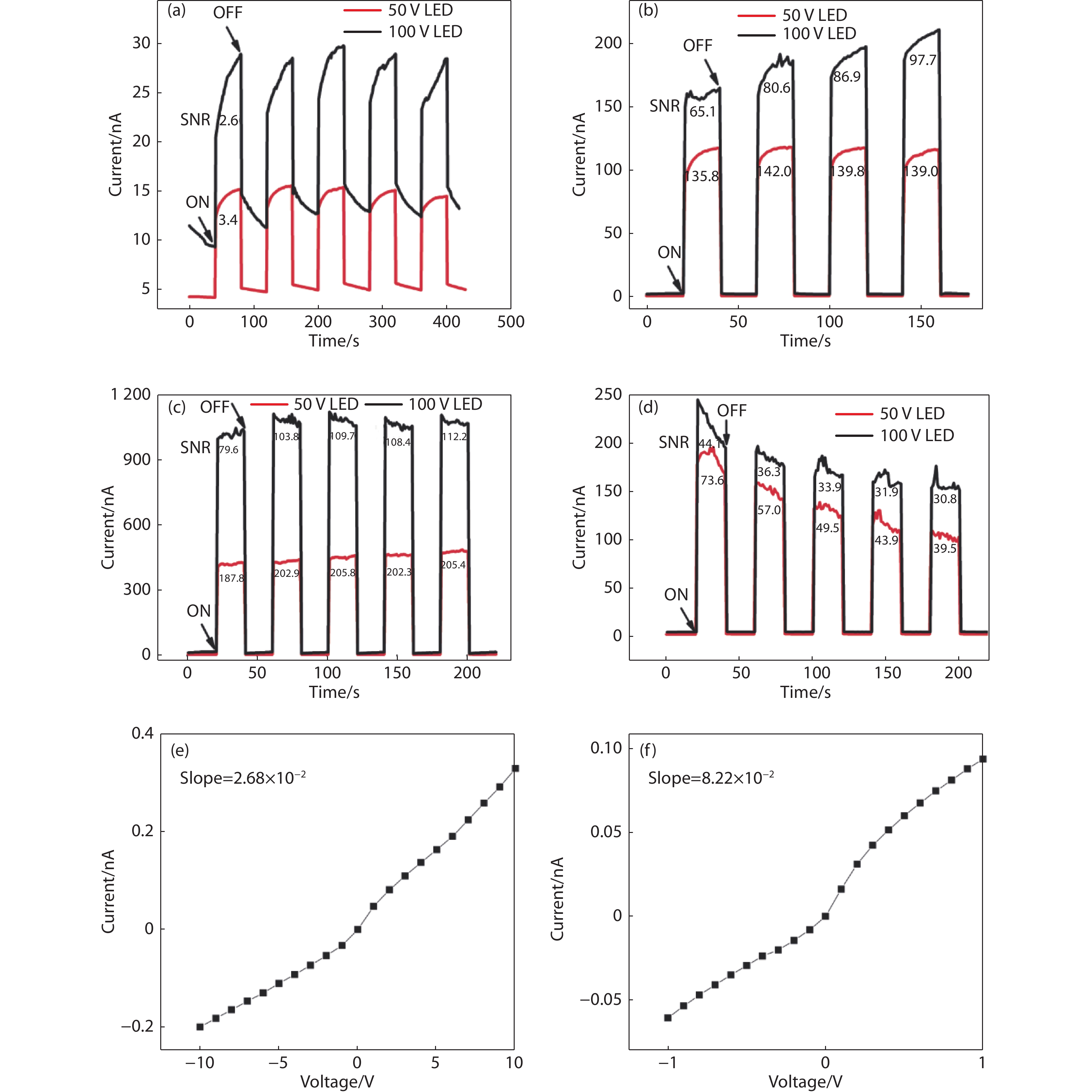
Figure 8.
Photoresponse results of CsPbBr3 detectors from different samples with Ag/Ag contact. (a) sample 1100. (b) sample 1127. (c) sample 1149. (d) sample 1153. Current-voltage (I-V) curves of (e) sample
1127 from -10 to 10 V, (f) sample1149 from - 1 to 1 V. -
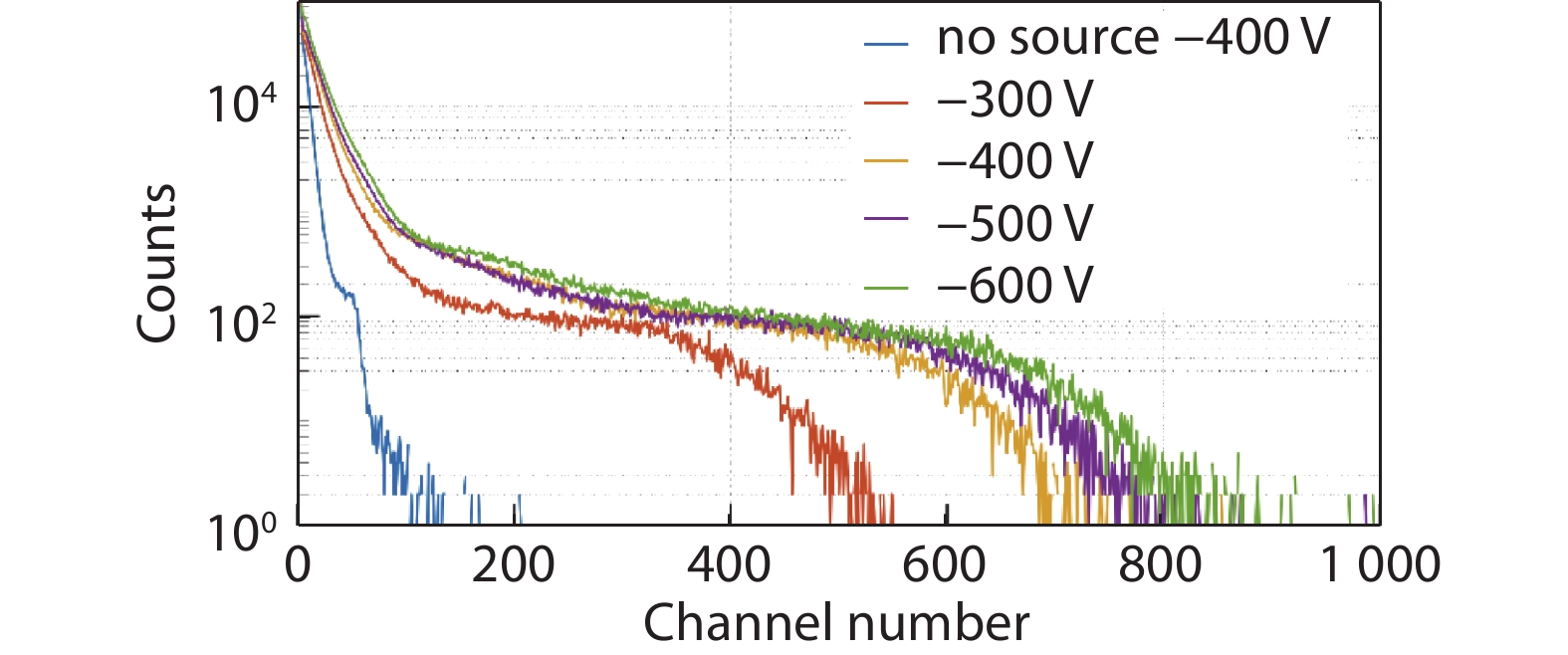
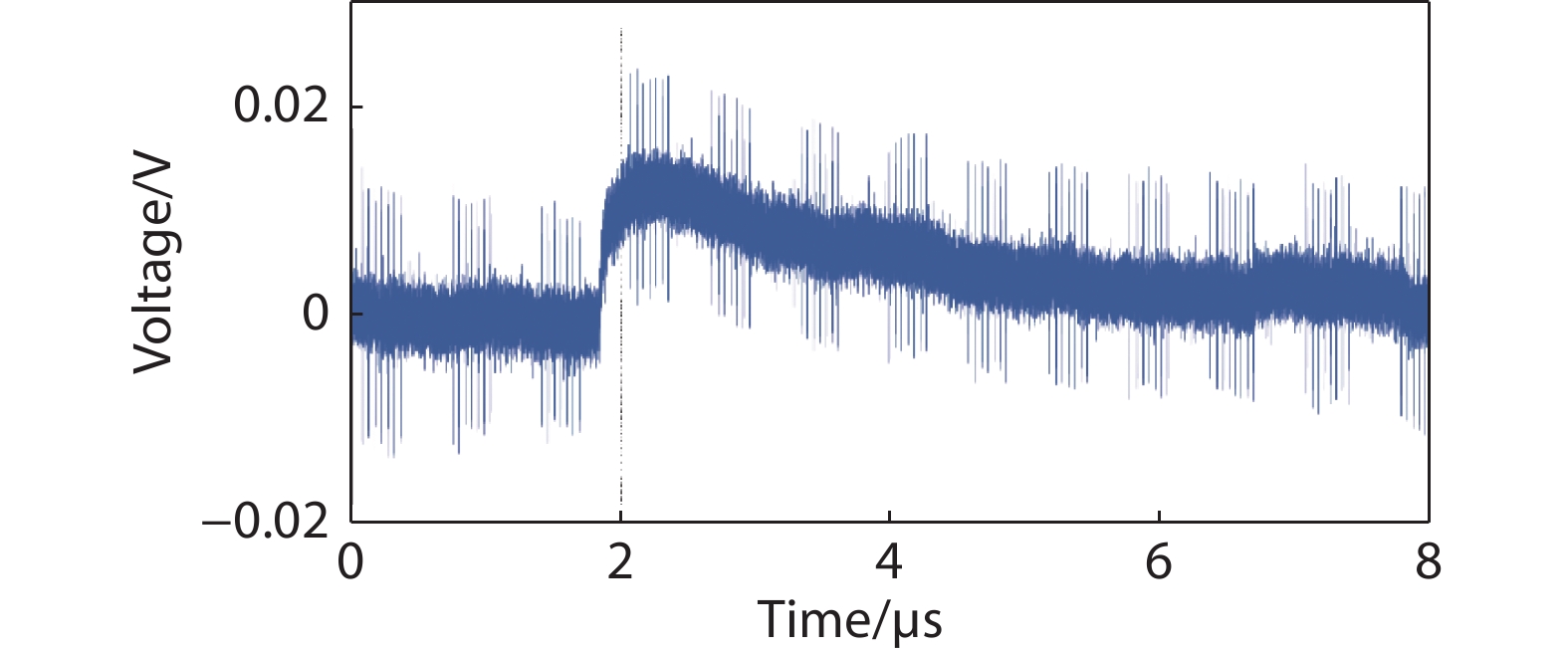
Figure 9.
Pulse-height spectrum collected for the CsPbBr3 crystal device with a Ga\CsPbBr3\Ga configuration.

 DownLoad:
DownLoad: