| Citation: | Qixian Zhang, Huicong Liu, Weiping Li, Haining Chen. Carbon-Based CsPbI3 Perovskite Solar Cells. Materials Lab 2022, 1, 220029. doi: 10.54227/mlab.20220029 |
Carbon-Based CsPbI3 Perovskite Solar Cells
Published as part of the Virtual Special Issue “Beihang University at 70”
-
Abstract
The perovskite solar cells (PSCs) based on carbon electrode (C-PSCs) are expected to address the instability issues faced by conventional PSCs. Recently, inorganic perovskites have been widely used as the light absorber in C-PSCs, which tended to further enhance device stability. Among various inorganic perovskites, CsPbI3 perovskite has been showing the greatest promise due to its suitable band gap (~1.7 eV) and high chemical stability. Benefiting from the progresses on phase stability, crystal quality and surface defect passivation, CsPbI3 C-PSCs have achieved the efficiency of over 15% and exhibited considerable enhancement in device stability. In this perspective, the main advances on CsPbI3 C-PSCs will be highlighted and the future research directions will be proposed.
-
Keywords:
- Perovskite solar cells /
- Inorganic perovskites /
- CsPbI3 /
- Carbon electrode.
-
-
References
1. M. Kim, J. Jeong, H. Lu, K. Lee Tae, T. Eickemeyer Felix, Y. Liu, W. Choi In, J. Choi Seung, Y. Jo, H.-B. Kim, S.-I. Mo, Y.-K. Kim, H. Lee, G. An Na, S. Cho, R. Tress Wolfgang, M. Zakeeruddin Shaik, A. Hagfeldt, Y. Kim Jin, M. Grätzel and S. Kim Dong, Science, 2022, 375, 302 2. X. Luo, Z. Shen, Y. Shen, Z. Su, X. Gao, Y. Wang, Q. Han and L. Han, Adv. Mater., 2022, 34, 2202100 3. J. Y. Seo, S. Akin, M. Zalibera, M. A. R. Preciado, H. S. Kim, S. M. Zakeeruddin, J. V. Milić and M. Grätzel, Advanced Functional Materials, 2021, 31, 2102124 4. C. Ding, R. Huang, C. Ahläng, J. Lin, L. Zhang, D. Zhang, Q. Luo, F. Li, R. Österbacka and C.-Q. Ma, Journal of Materials Chemistry A, 2021, 9, 7575 5. H. Chen and S. Yang, Adv. Mater., 2017, 29, 1603994 6. H. Chen, S. Xiang, W. Li, H. Liu, L. Zhu and S. Yang, Solar RRL, 2018, 2, 1700188 7. X. Chang, W. Li, L. Zhu, H. Liu, H. Geng, S. Xiang, J. Liu and H. Chen, ACS Applied Materials & Interfaces, 2016, 8, 33649 8. J. Liang, C. Wang, Y. Wang, Z. Xu, Z. Lu, Y. Ma, H. Zhu, Y. Hu, C. Xiao, X. Yi, G. Zhu, H. Lv, L. Ma, T. Chen, Z. Tie, Z. Jin and J. Liu, Journal of the American Chemical Society, 2016, 138, 15829 9. Q. Zhou, J. Duan, J. Du, Q. Guo, Q. Zhang, X. Yang, Y. Duan and Q. Tang, Adv. Sci., 2021, 8, 2101418 10. C. Dong, B. Xu, D. Liu, E. G. Moloney, F. Tan, G. Yue, R. Liu, D. Zhang, W. Zhang and M. I. Saidaminov, Materials Today, 2021, 50, 239 11. Q. Guo, J. Duan, J. Zhang, Q. Zhang, Y. Duan, X. Yang, B. He, Y. Zhao and Q. Tang, Adv. Mater., 2022, 34, 2202301 12. W. Zhu, J. Ma, W. Chai, T. Han, D. Chen, X. Xie, G. Liu, P. Dong, H. Xi, D. Chen, J. Zhang, C. Zhang and Y. Hao, Solar RRL, 2022, 6, 2200020 13. H. Wang, H. Liu, Z. Dong, T. Song, W. Li, L. Zhu, Y. Bai and H. Chen, Nano Energy, 2021, 89, 106411 14. X. Fu, W. Li, X. Zeng, C. Yan, X. Peng, Y. Gao, Q. Wang, J. Cao, S. Yang and W. Yang, J. Phys. Chem. Lett., 2022, 13, 2217 15. H. Wang, H. Liu, Z. Dong, W. Li, L. Zhu and H. Chen, Nano Energy, 2021, 84, 105881 16. W. Meng, Y. Hou, A. Karl, E. Gu, X. Tang, A. Osvet, K. Zhang, Y. Zhao, X. Du, J. Garcia Cerrillo, N. Li and C. J. Brabec, ACS Energy Letters, 2020, 5, 271 17. Z. Li, F. Zhou, Q. Wang, L. Ding and Z. Jin, Nano Energy, 2020, 71, 104634 18. J. Liang, C. Wang, P. Zhao, Z. Lu, Y. Ma, Z. Xu, Y. Wang, H. Zhu, Y. Hu, G. Zhu, L. Ma, T. Chen, Z. Tie, J. Liu and Z. Jin, Nanoscale, 2017, 9, 11841 19. S. Xiang, W. Li, Y. Wei, J. Liu, H. Liu, L. Zhu and H. Chen, Nanoscale, 2018, 10, 9996 20. S. Xiang, Z. Fu, W. Li, Y. Wei, J. Liu, H. Liu, L. Zhu, R. Zhang and H. Chen, ACS Energy Letters, 2018, 3, 1824 21. J. Liang, X. Han, J. H. Yang, B. Zhang, Q. Fang, J. Zhang, Q. Ai, M. M. Ogle, T. Terlier, A. A. Marti and J. Lou, Adv. Mater., 2019, 31, 1903448 22. S. Xiang, W. Li, Y. Wei, J. Liu, H. Liu, L. Zhu, S. Yang and H. Chen, iScience, 2019, 15, 156 23. H. Wang, H. Liu, Z. Dong, X. Wei, Y. Song, W. Li, L. Zhu, Y. Bai and H. Chen, Nano Energy, 2022, 94, 106925 24. S. Tan, B. Yu, Y. Cui, F. Meng, C. Huang, Y. Li, Z. Chen, H. Wu, J. Shi, Y. Luo, D. Li and Q. Meng, Angew. Chem. Int. Ed. Engl., 2022, 61, e202201300 25. X. Wang, Y. Wang, Y. Chen, X. Liu and Y. Zhao, Adv. Mater., 2021, 33, 2103688 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
Figure 1.
The device structure of (a) conventional PSCs and (b) PSCs based on carbon electrodes (C-PSCs). (c) Energy band diagrams of CsPbI3-xBrx C-PSCs[10]. Copyright 2021, Elsevier Ltd. (d) Evolution of the reported PCEs of CsPbI3 C-PSCs[13,15,18-23].

 Qixian Zhang is a doctoral student of the School of Materials Science and Engineering, Beihang university. He is pursuing a Ph.D. degree under the supervision of Prof. Haining Chen. His current research interest focuses on carbon-based perovskite solar cells.
Qixian Zhang is a doctoral student of the School of Materials Science and Engineering, Beihang university. He is pursuing a Ph.D. degree under the supervision of Prof. Haining Chen. His current research interest focuses on carbon-based perovskite solar cells.  Haining Chen is currently an associate professor at the School of Materials Science and Engineering, Beihang University. He obtained his Ph.D. degree from Beihang University in 2013 following which he worked as a postdoctoral fellow at The Hong Kong University of Science and Technology (Prof. Shihe Yang's group). His recent research interests include perovskite solar cells and water splitting.
Haining Chen is currently an associate professor at the School of Materials Science and Engineering, Beihang University. He obtained his Ph.D. degree from Beihang University in 2013 following which he worked as a postdoctoral fellow at The Hong Kong University of Science and Technology (Prof. Shihe Yang's group). His recent research interests include perovskite solar cells and water splitting. 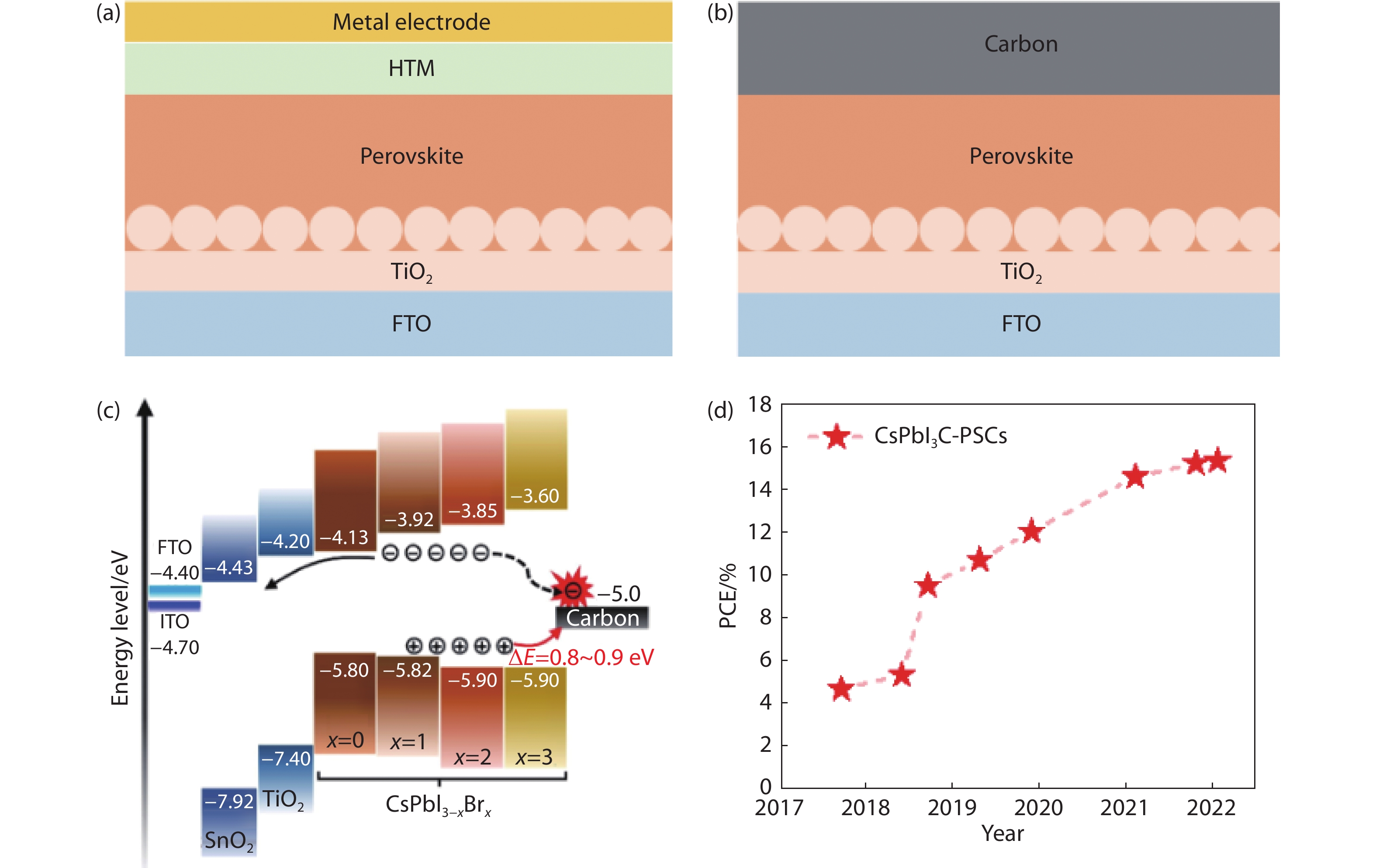

 DownLoad:
DownLoad:

