| Citation: | Haitao Li, Mingyang Li, Xiaoxue Jiang, Changda Zhu, Hongguang Wang, Aowei Deng, Guozhong Zang, Yongjun Gu. Enhanced piezoelectric properties for potassium sodium niobate lead-free piezoelectric ceramics prepared by microwave technology[J]. Materials Lab, 2024, 3(1): 230019. doi: 10.54227/mlab.20230019 |
Enhanced piezoelectric properties for potassium sodium niobate lead-free piezoelectric ceramics prepared by microwave technology
-
Abstract
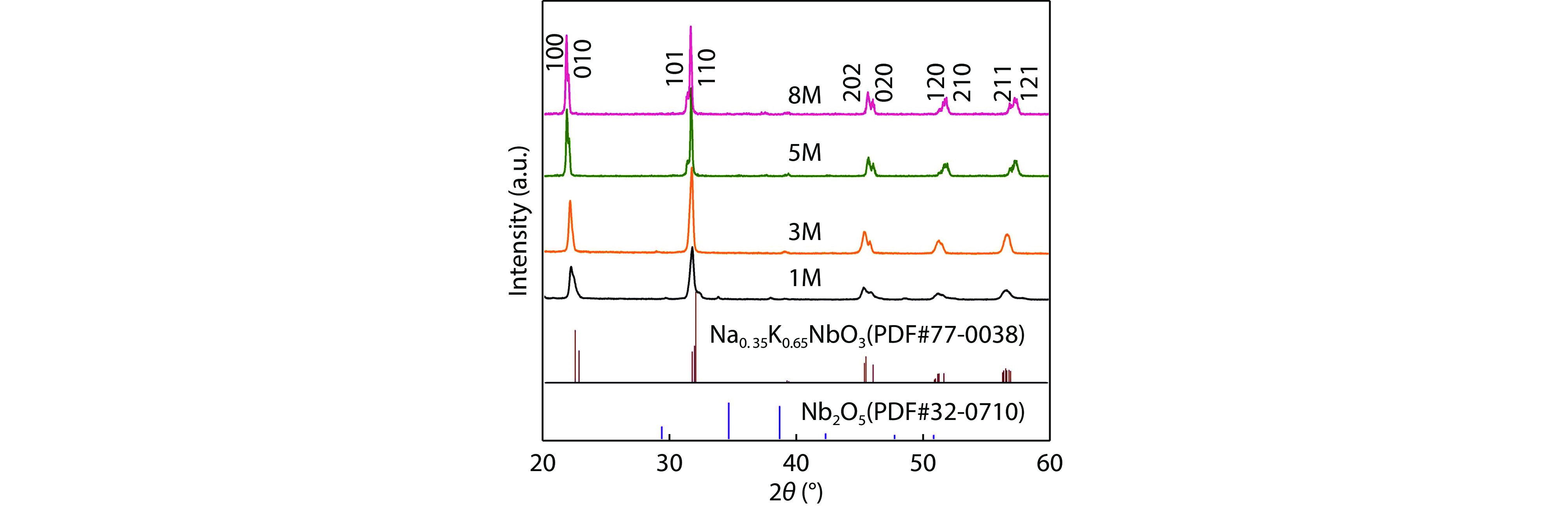
Potassium-sodium niobate (Na, K)NbO3 (NKN) powder was synthesized at low temperature via microwave-assisted hydrothermal solovthermal method (MHSM). The resultant powder was characterized by X-ray diffraction (XRD) and scanning electron microscope (SEM). XRD results showed that pure (Na, K)NbO3 powder with a single perovskite structure was successfully synthesized when the concentration of mineralization exceeded 1 M. In order to reduce the volatilization of alkaline elements, NKN ceramics derived from 5M-powder were prepared in a microwave furnace. Microstructure, stoichiometry, and electrical properties of the obtained ceramics were investigated. The piezoelectric coefficient (
d 33), electromechanical coupling coefficient (k p) and remnant polarization (P r) of the sample sintered at1050 °C show optimal values of 132 pC/N, 38% and 26.3 µC cm-2, respectively. The results indicate that the microwave heating is a promising method for synthesizing and sintering NKN-based ceramics. -
-
References
1. Y. Saito, H. Takao, T. Tani, T. Nonoyama, K. Takatori, T. Homma, T. Nagaya, M. Nakamura, Nature, 2004, 432, 84 2. T. Q. Shao, H. L. Du, H. Ma, S. Qu, J. Wang, J. Wang, X. Wei, Z. Xu, J. Mater. Chem. A, 2017, 5, 554 3. H. T. Li, Q. Cao, F. Wang, M. H. Zhang, Q. Yu, R. Y. Dong, J. Alloys Comp., 2015, 634, 163 4. J. Ma, B. Wu, W. J. Wu, J. Mater. Sci.-Mater. Electron., 2017, 28, 4458 5. H. T. Li, B. P. Zhang, Q. Zhang, P. P. Shang, G. L. Zhao, Int. J. Miner. Metall. Mater., 2010, 17, 340 6. H. T. Li, B. P. Zhang, J. B. Wen, R. H. Xu, Q. Li, J. Inorg. Mater., 2012, 27, 385 7. S. R. Kim, J. H. Yoo, J. H. Kim, Y. S. Cho, J. W. Park, Nano Energy, 2021, 79, 105445 8. Y. W. Liu, Y. P. Pu, Q. Jin, Sci. Adv. Mater., 2018, 10, 257 9. L. Wu, J. L. Zhang, C. L. Wang, J. C. Li, J. Appl. Phys., 2008, 103, 084116 10. Y. L. Su, X. M. Chen, Z. D. Yu, J. Mater. Sci., 2017, 52, 2934 11. B. P. Zhang, J. F. Li, K. Wang, H. L. Zhang, J. Am. Chem. Soc., 2006, 89, 1605 12. H. T. Li, B. P. Zhang, M. Cui, W. G. Yang, N. Ma, J. F. Li, Curr. Appl. Phys., 2011, 11, 184 13. H. T. Li, Q. Li, Y. F. Yan, R. H. Xu, J. Inorg. Mater., 2015, 30, 369 14. Y. Lu, H. T. Li, Q. Li, B. P. Zhang, Rare metals, 2010, 29, 243 15. H. Y. Park, C. W. Ahn, K. H. Cho, S. Nahm, H. G. Lee, H. W. Kang, D. H. Kim, K. S. Park, J. Am. Ceram. Soc., 2007, 90, 4066 16. M. R. Bafandeh, J. S. Lee, H. S. Han, J. Electroceram., 2014, 33, 128 17. A. Chowdhury, J. Bould, Y. Zhang, C. James, S. J. Milne, J. Nanopart. Res., 2010, 12, 209 18. C. Pithan, Y. Shiratori, J. Dornseiffer, F. H. Haegel, A. Magrez, R. Waser, J. Cryst. Growth, 2005, 280, 191 19. H. Gu, K. Zhu, X. Pang, B. Shao, J. Qiu, H. Ji, Ceram. Int., 2012, 38, 1807 20. L. Bai, K. Zhu, L. Su, J. Qiu, H. Ji, Mater. Lett., 2010, 64, 77 21. H. T. Li, Y. F. Yan, G. X. Wang, Q. Li, Y. J. Gu, J. Mater. Sci: Mater. Electron, 2018, 29, 746 22. Y. Zhou, J. L. Yu, M. Guo, M. Zhang, Ferroelectrics, 2010, 404, 69 23. M. Feizpour, H. Barzegar Bafrooei, R. Hayati, T. Ebadzadeh, Ceram. Int., 2014, 40, 871 24. R. López-Juárez, R. Castañeda-Guzmán, M. E. Villafuerte-Castrejón, Ceram. Int., 2014, 40, 14757 25. K. Wang, J. F. Li, N. Liu, Appl. Phys. Lett., 2008, 93, 092904 26. L. Jin, F. Li, S. J. Zhang, J. Am. Chem. Soc., 2014, 97, 1 27. R. Zuo, J. Rodel, R. Chen, L. Li, J. Am. Ceram. Soc., 2006, 89, 2010 28. H. Evelyn, D. Matthew, D. Dragan, S. Nava, Appl. Phys. Lett., 2005, 87, 182905 29. P. Kumar, P. Palei, Ceram. Int., 2010, 36, 1725 30. H. Birol, D. Damjanovic, N. Setter, J. Eur. Ceram. Soc., 2006, 26, 861 31. P. Zhao, B.P. Zhang, J. F. Li, Appl. Phys. Lett., 2007, 90, 242909 32. H. T. Li, B. P. Zhang, P. P. Shang, Y. Fan, Q. Zhang, J. Am. Ceram. Soc., 2011, 94, 628 33. V. J. Tennery, K. W. Hang, J. Appl. Phys., 1968, 39, 4749 34. P. Li, Y. Huan, W. W. Yang, Acta Mater., 2019, 165, 486 35. D. K. Liu, X. C. Zhang, W. B. Su, J. Alloy. Compd., 2019, 779, 800 36. X. Y. Gao, Z. X. Cheng, Z. B. Chen, Nat. Commun., 2021, 12, 881 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
Figure 1.
XRD patterns of NKN powders synthesized by MHSM with different mineralizer concentrations.
-
Figure 2.
SEM images of NKN powders with different mineralizer concentrations: a 3 M; b 5 M; c 8 M.
-
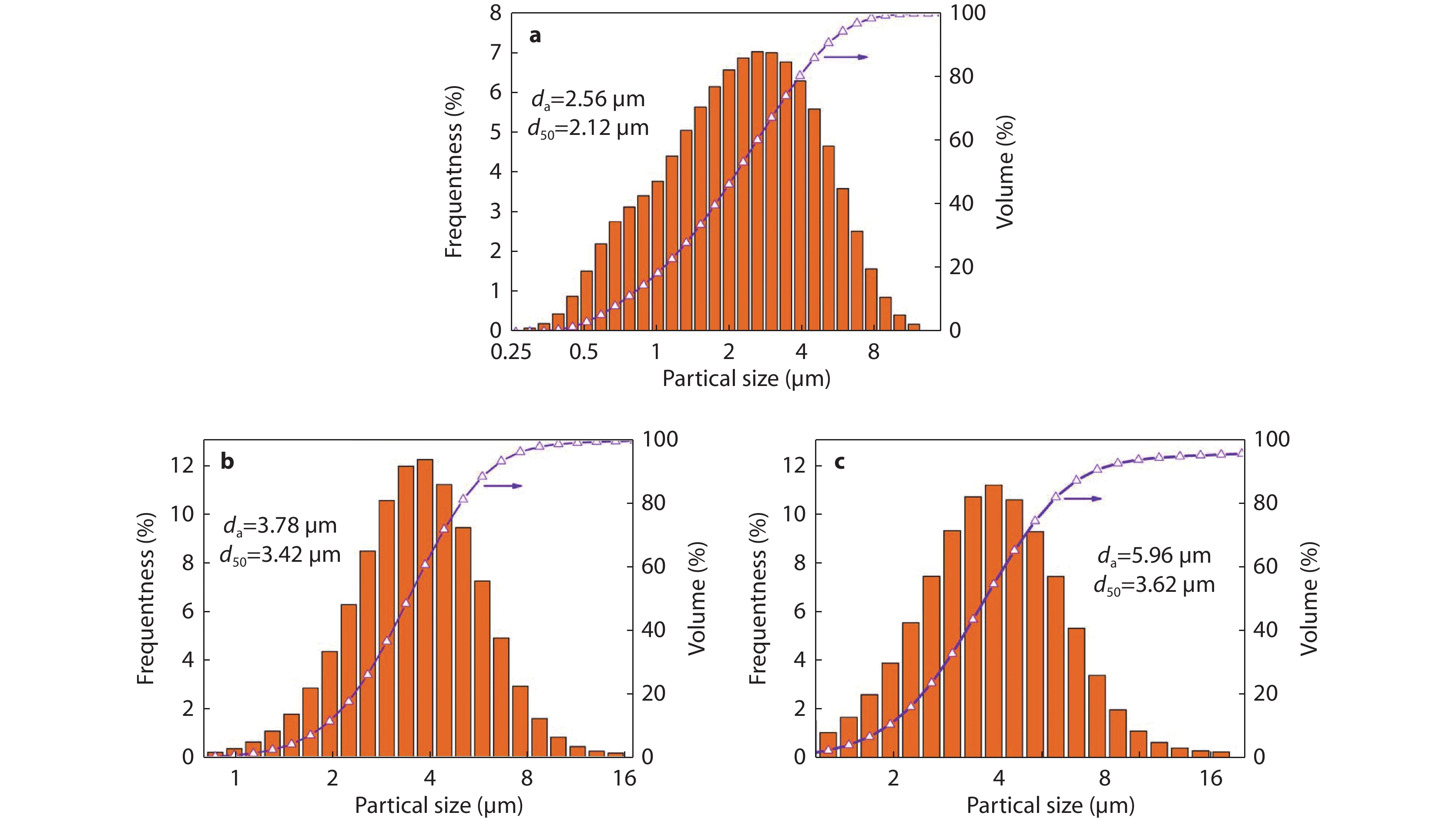
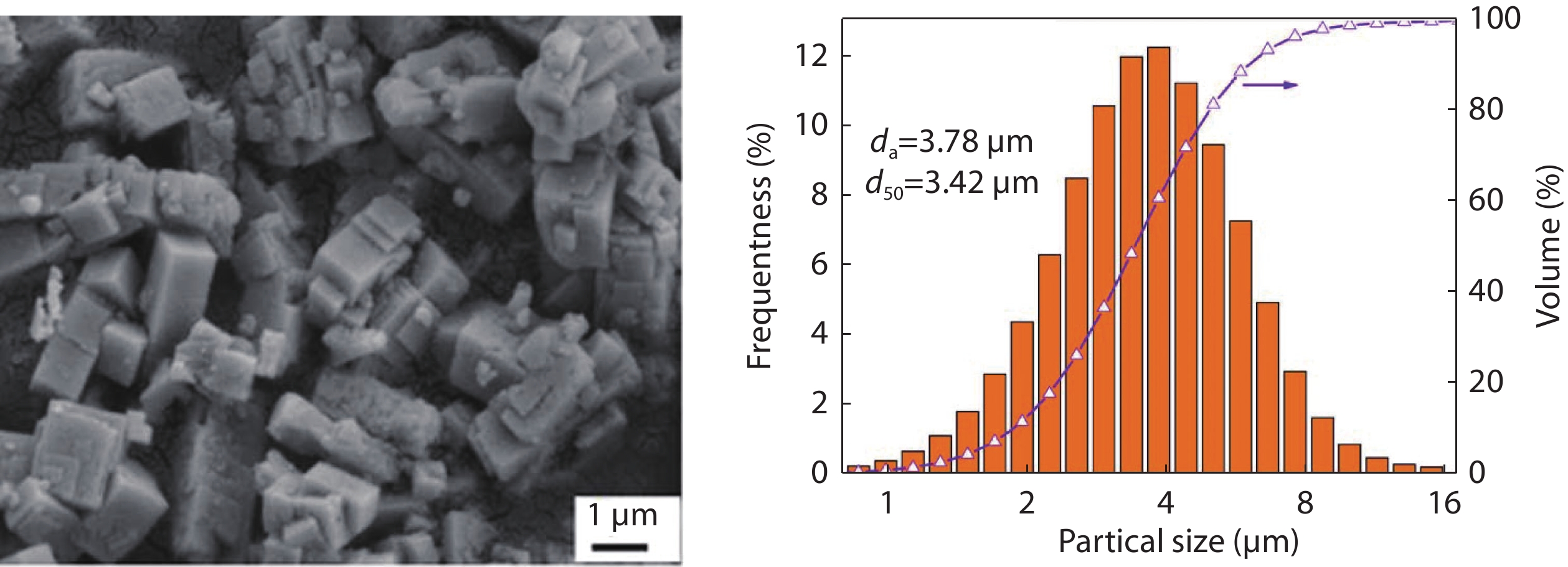
Figure 3.
Particle size distribution of NKN powders with different mineralizer concentrations: a 3 M; b 5 M; c 8 M.
-
Figure 4.
SEM images of the fracture surface of NKN ceramics sintered at different temperatures: a
1000 °C; b1020 °C; c1050 °C; d1080 °C. -
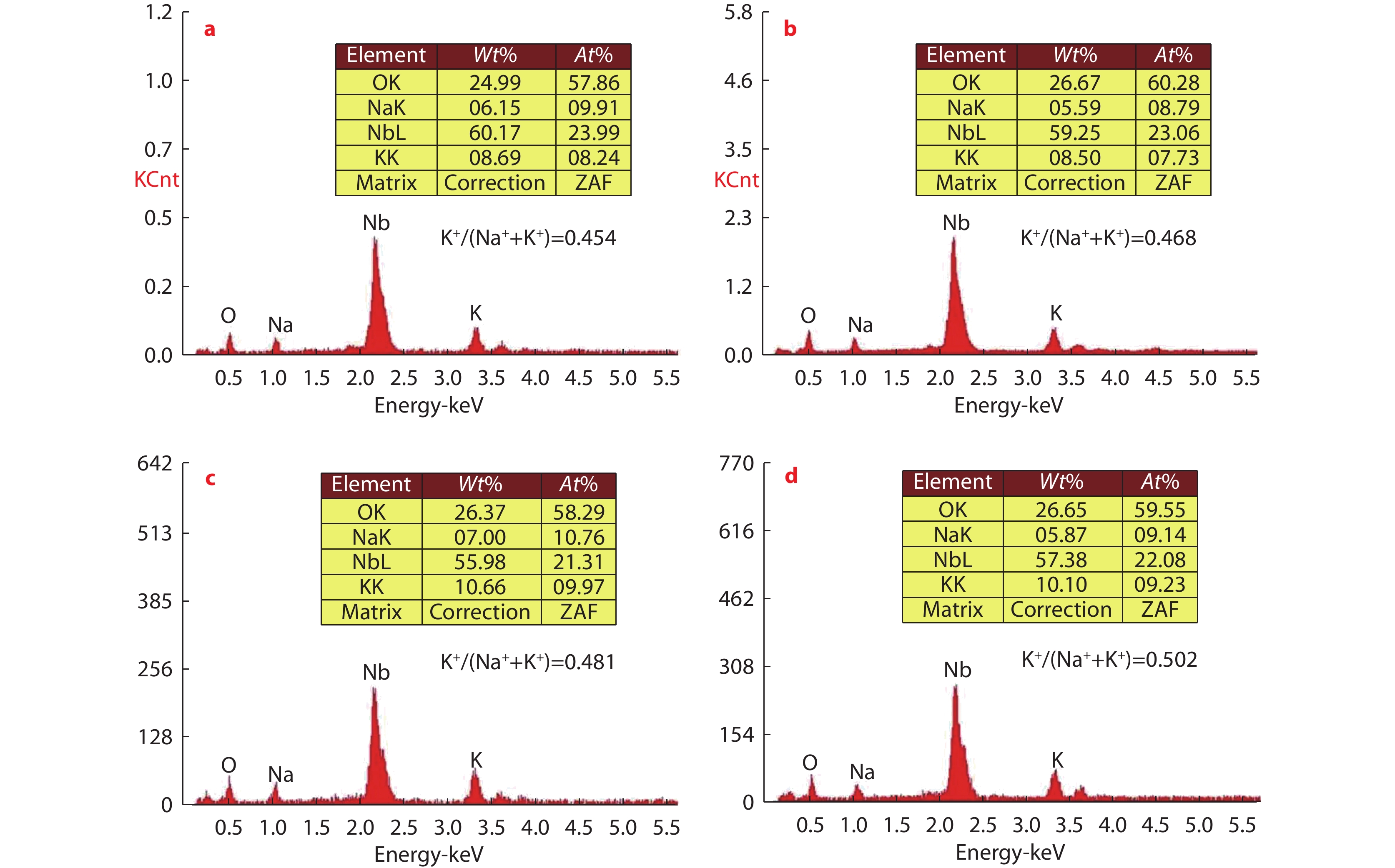
Figure 5.
EDS spectra of NKN ceramics sintered at different temperatures: a
1000 °C; b1020 °C; c1050 °C; d1080 °C. -
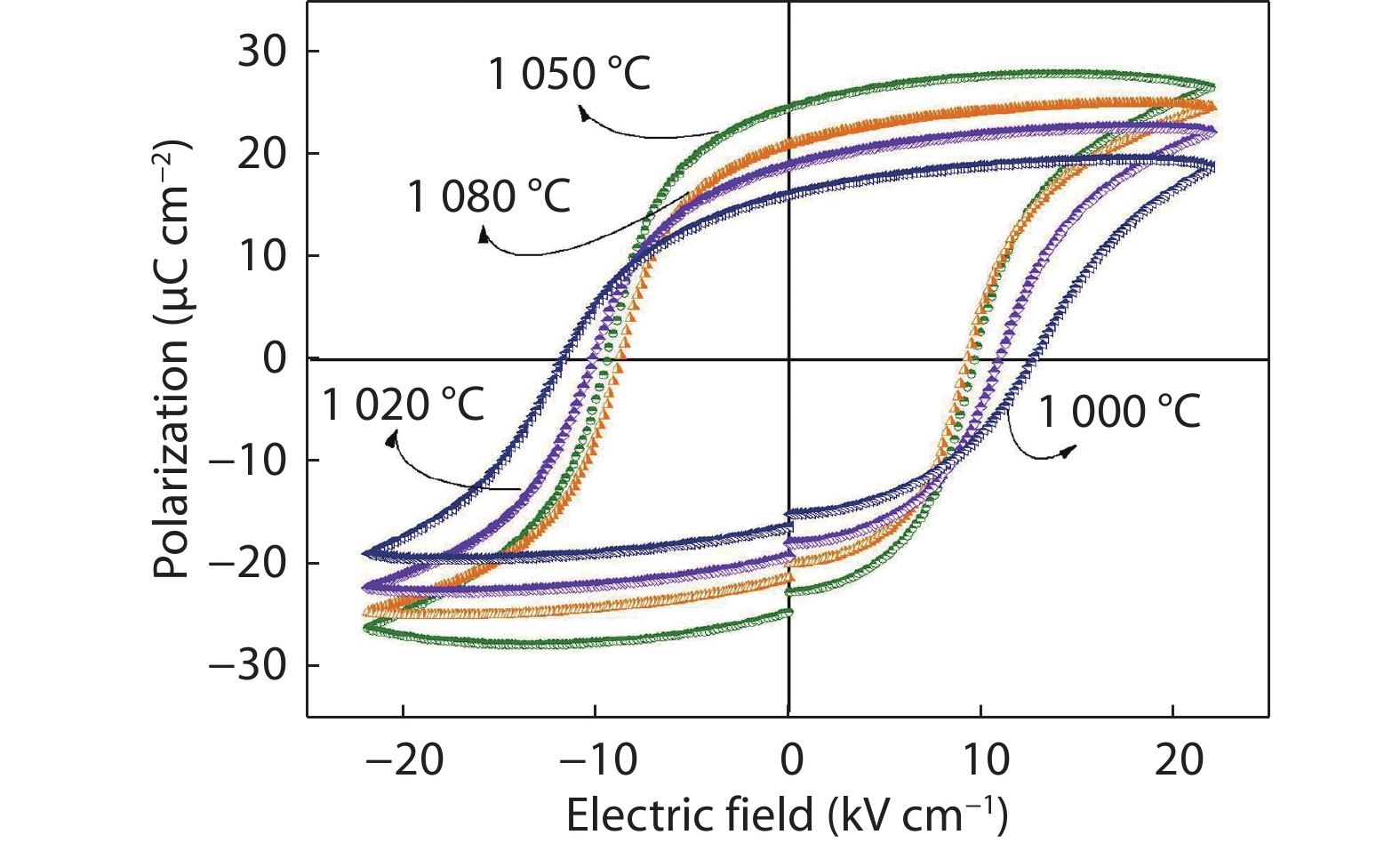
Figure 6.
P-E hysteresis loops of NKN ceramics sintered at different temperatures.

 DownLoad:
DownLoad: