| Citation: | Xiyu Wang, Lingling Zhang, Zijian Wang, Xiao Wang, Shuyan Song, Hongjie Zhang. Recent strategies for switching the catalytic selectivity in CO2 hydrogenation[J]. Materials Lab, 2024, 3(3): 240004. doi: 10.54227/mlab.20240004 |
Recent strategies for switching the catalytic selectivity in CO2 hydrogenation
-
Abstract
Thermal catalysis is vital for converting carbon dioxide during hydrogenation into high-value-added chemicals. However, the intricate network of reaction pathways and intermediates leads to a diverse array of CO2 hydrogenation products, making achieving high selectivity difficult. Furthermore, in practical applications, changes to the catalyst induced by various factors can significantly alter its properties, resulting in a sudden shift in product selectivity, often referred to as selectivity reversal. Therefore, investigating the factors contributing to this selectivity reversal is essential for establishing a theoretical basis for regulating selectivity in CO2 hydrogenation. This paper reviews the factors influencing selectivity reversal, such as heteroatom modification, metal-support interactions (MSI), changes in the size of the active component, and intermetallic electronic interactions. Additionally, strategies to modulate the selectivity of CO2 hydrogenation are discussed. In conclusion, by exploring catalyst selectivity reversal and presenting methods to manipulate the selectivity of CO2 hydrogenation, this paper offers a guiding framework for the targeted design of catalysts and the precise modulation of CO2 hydrogenation products.
-
Keywords:
- CO2 hydrogenation /
- Selectivity /
- Catalyst /
- Nanomaterials /
- Mechanization
-
-
References
1. J. Hu, Y. Cai, J. Xie, D. Hou, L. Yu, D. Deng, Chem, 2024, 10, 1084 2. M. Z. Chishti, S. Ullah, I. Ozturk, A. Usman, Environ. Sci. Pollut. Res., 2020, 27, 27721 3. M. Tawalbeh, M. Al-Ismaily, B. Kruczek, F. H. Tezel, Chemosphere, 2021, 263, 127935 4. J. Yang, W. Cai, M. Ma, L. Li, C. Liu, X. Ma, L. Li, X. Chen, Sci. Total Environ., 2020, 711, 134569 5. R. Ahmed, G. Liu, B. Yousaf, Q. Abbas, H. Ullah, M. U. Ali, J. Cleaner Prod., 2020, 242, 118409 6. T. R. Knutson, R. E. Tuleya, J. Clim., 2004, 17, 3477 7. X. T. Wu, X. W, L. L. Zhang, X. M. Wang, S. Y. Song, H. J. Zhang, Angew. Chem. Int. Ed, 2024, 63, e202317594 8. H. Wang, M. Bootharaju, J. Kim, Y. Wang, K. Wang, M. Zhao, R. Zhang, J. Xu, T. Hyeon, X. Wang, S. Song, H. Zhang, J. Am. Chen. Soc., 2023, 145, 2264 9. Z. Wang, W. Huang, H. Wang, J. Gao, R. Zhang, G. Xu, Z. Wang, J. Cleaner Prod., 2024, 434, 140365 10. T. Li, X. Meng, F. Jia, L. Chen, L. Wang, J. Cleaner Prod., 2023, 418, 138115 11. D. Patnaik, A. K. Pattanaik, D. K. Bagal, A. Rath, Int. J. Hydrogen Energy, 2023, 48, 23449 12. A. Bhavsar, D. Hingar, S. Ostwal, I. Thakkar, S. Jadeja, M. Shah, Case Stud. Chem. Environ. Eng., 2023, 8, 100368 13. A. S. Varling, T. H. Christensen, V. Bisinella, Waste Management, 2023, 157, 168 14. B. Dziejarski, R. Krzyżyńska, K. Andersson, Fuel, 2023, 342, 127776 15. S. Nagireddi, J. R. Agarwal, D. Vedapuri, ACS Eng. Au, 2024, 4, 22 16. L. Fu, Z. Ren, W. Si, Q. Ma, W. Huang, K. Liao, Z. Huang, Y. Wang, J. Li, P. Xu, J. CO2 Util., 2022, 66, 102260 17. A. Yagmur Goren, D. Erdemir, I. Dincer, Environ. Res., 2024, 240, 117503 18. H. A. Baalbaki, H. Roshandel, J. E. Hein, P. Mehrkhodavandi, Catal. Sci. Technol., 2021, 11, 2119 19. G. Hu, Z. Li, D. Ma, C. Ye, L. Zhang, C. Gao, L. Liu, X. Chen, Nat. Catal., 2021, 4, 395 20. J. Godin, W. Liu, S. Ren, C. C. Xu, J. Environ. Chem. Eng., 2021, 9, 105644 21. W. Fei, D. David, Minerals and Mineral Materials, 2022, 1, 4 22. S. Schlager, A. Fuchsbauer, M. Haberbauer, H. Neugebauer, N. S. Sariciftci, J. Mater. Chem. A, 2017, 5, 2429 23. P. Tcvetkov, A. Cherepovitsyn, S. Fedoseev, Sustainability, 2019, 11, 5834 24. S. Fang, M. Rahaman, J. Bharti, E. Reisner, M. Robert, G. A. Ozin, Y. H. Hu, Nat. Rev. Methods Primers, 2023, 3, 61 25. W. Jiang, H. Loh, B. Q. L. Low, H. Zhu, J. Low, J. Z. X. Heng, K. Y. Tang, Z. Li, X. J. Loh, E. Ye, Y. Xiong, Appl. Catal. B, 2023, 321, 122079 26. T. Lu, T. Xu, S. Zhu, J. Li, J. Wang, H. Jin, X. Wang, J. J. Lv, Z. J. Wang, S. Wang, Adv. Mater., 2023, 35, 2310433 27. Z.-H. He, C.-C. Li, S.-Y. Yang, J. Liu, H.-H. Cao, K. Wang, W. Wang, Y. Yang, Z.-T. Liu, Catal. Sci. Technol., 2023, 13, 6675 28. H. Cao, Y. Li, Q. Hu, J. Wu, L. Ji, J. Mater. Chem. A, 2023, 11, 19645 29. J. Gao, S. C. S. Shiong, Y. Liu, Chem. Eng. J., 2023, 145033 30. Y. He, L. Yin, N. Yuan, G. Zhang, Chem. Eng. J., 2024, 481, 148754 31. G. Liao, G. Ding, B. Yang, C. Li, Precis. Chem., 2024, 2, 49 32. D. Yang, X. Wang, SmartMat, 2022, 3, 54 33. J. Lin, Y. Zhang, P. Xu, L. Chen, Mater. Rep.: Energy, 2023, 3, 100194 34. X. She, Y. Wang, H. Xu, S. Chi Edman Tsang, S. Ping Lau, Angew. Chem. Int. Ed. Engl., 2022, 61, e202211396 35. J. Gao, S. Choo Sze Shiong, Y. Liu, Chem. Eng. J., 2023, 472, 145033 36. S. Roy, A. Cherevotan, S. C. Peter, ACS Energy Lett., 2018, 3, 1938 37. N. Yusuf, F. Almomani, H. Qiblawey, Fuel, 2023, 345, 128178 38. N. S. Featherstone, E. van Steen, Catal. Today, 2023, 423, 113951 39. Y. Wang, L. R. Winter, J. G. Chen, B. Yan, Green Chem., 2021, 23, 249 40. W. J. Lee, C. Li, H. Prajitno, J. Yoo, J. Patel, Y. Yang, S. Lim, Catal. Today, 2021, 368, 2 41. P. H. Yan, H. Peng, J. Vogrin, H. Rabiee, Z. H. Zhu, J. Mater. Chem. A, 2023, 11, 17938 42. T. Fan, H. Liu, S. Shao, Y. Gong, G. Li, Z. Tang, J. Phys. Chem. Lett., 2021, 12, 10486 43. C. Yin, Q. Li, J. Zheng, Y. Ni, H. Wu, A.-L. Kjøniksen, C. Liu, Y. Lei, Y. Zhang, Advanced Powder Materials, 2022, 1, 100055 44. C. Han, Y. Gao, L. Qin, Y. Cao, S. Wang, J. Li, Sep. Purif. Technol., 2025, 354, 128852 45. Y. Jiang, J. Lang, X. Wu, Y. H. Hu, Catal. Today, 2020, 356, 570 46. R. Yao, B. Wu, Y. Yu, N. Liu, Q. Niu, C. Li, J. Wei, Q. Ge, Appl. Catal. B-Environ., 2024, 355, 124159 47. F. Lin, X. Jiang, N. Boreriboon, Z. Wang, C. Song, K. Cen, Appl. Catal. A, 2019, 585, 117210 48. A. Parastaev, V. Muravev, E. Huertas Osta, A. J. van Hoof, T. F. Kimpel, N. Kosinov, E. J. Hensen, Nat. Catal., 2020, 3, 526 49. Y. Xie, J. Wen, Z. Li, J. Chen, Q. Zhang, P. Ning, J. Hao, ACS Mater. Lett., 2023, 5, 2629 50. C. Zhang, L. Wang, Y. Chen, X. He, Y. Song, O. M. Gazit, Z. Zhong, Chem. Eng. J., 2023, 475, 146102 51. C. Meng, G. Zhao, X.-R. Shi, Q. Nie, Y. Liu, Y. Lu, Appl. Catal. B, 2022, 316, 121699 52. B. Yang, Y. Wang, L. Li, B. Gao, L. Zhang, L. Guo, Catal. Sci. Technol., 2022, 12, 1159 53. C. Meng, G. Zhao, X.-R. Shi, P. Chen, Y. Liu, Y. Lu, Sci. Adv., 2021, 7, eabi6012 54. X. Wang, H. Zhang, H. Qin, K. Wu, K. Wang, J. Ma, W. Fan, Fuel, 2023, 346, 128381 55. Y. Yang, X. Zhu, L. Wang, J. Lang, G. Yao, T. Qin, Z. Ren, L. Chen, X. Liu, W. Li, Y. Wan, Nat. Commun., 2022, 13, 2754 56. X. Zhang, M. Zhang, Y. Deng, M. Xu, L. Artiglia, W. Wen, R. Gao, B. Chen, S. Yao, X. Zhang, M. Peng, J. Yan, A. Li, Z. Jiang, X. Gao, S. Cao, C. Yang, A. J. Kropf, J. Shi, J. Xie, M. Bi, J. A. van Bokhoven, Y. -W. Li, X. Wen, M. Flytzani-Stephanopoulos, C. Shi, W. Zhou, D. Ma, Nature, 2021, 589, 396 57. T.-N. Ye, S.-W. Park, Y. Lu, J. Li, M. Sasase, M. Kitano, T. Tada, H. Hosono, Nature, 2020, 583, 391 58. T. He, W. Wang, F. Shi, X. Yang, X. Li, J. Wu, Y. Yin, M. Jin, Nature, 2021, 598, 76-81 59. J. M. Crawford, B. E. Petel, M. J. Rasmussen, T. Ludwig, E. M. Miller, S. Halingstad, S. A. Akhade, S. H. Pang, M. M. Yung, Appl. Catal. , A, 2023, 663, 119292 60. C. Wu, J. Shen, X. An, Z. Wu, S. Qian, S. Zhang, Z. Wang, B. Song, Y. Cheng, B. Yan, T.-K. Sham, X. Zhang, C. Li, K. Feng, L. He, ACS Catal., 2024, 14, 8592 61. S. Bao, T. Liu, H. Fu, Z. Xu, X. Qu, S. Zheng, D. Zhu, ACS Appl. Mater. Interfaces, 2023, 15, 45949 62. X. Wei, G. Johnson, Y. Ye, M. Cui, S.-W. Yu, Y. Ran, J. Cai, Z. Liu, X. Chen, W. Gao, P. J. L. Bean, W. Zhang, T. Y. Zhao, F. A. Perras, E. J. Crumlin, X. Zhang, R. J. Davis, Z. Wu, S. Zhang, J. Am. Chem. Soc., 2023, 145, 14298 63. J. Zhang, M. Gao, R. Wang, X. Li, T. Huang, J. Wang, Y. Wang, Z. Zheng, Nano Res., 2023, 16, 4786 64. J. Wang, K. Liu, J. Zhao, X. Li, B. Yin, B. Jiang, H. Li, RSC Adv., 2024, 14, 6502 65. M. Chen, L. G. Liu, X. Y. Chen, X. X. Qin, J. H. Zhang, S. H. Xie, F. D. Liu, H. He, C. B. Zhang, Nat. Commun., 2024, 15, 9478 66. F. Yang, T. Zhang, J. Zhao, J. Xiao, W. Zhou, J. Catal., 2024, 429, 115242 67. Y. Xie, J. Chen, J. Wen, Z. Li, F. Cao, S. Zhang, Q. Sun, P. Ning, Q. Zhang, J. Hao, ACS Catal., 2024, 14, 12214 68. S. Li, Y. Xu, H. Wang, B. Teng, Q. Liu, Q. Li, L. Xu, X. Liu, J. Lu, Angew. Chem. Int. Ed., 2023, 62, e202218167 69. M. Shi, G. Ren, Z. Zhang, Z. Li, X. Meng, Sol. Energy, 2023, 261, 33-42 70. F. Qi, J. Peng, Z. Liang, J. Guo, J. Liu, T. Fang, H. Mao, Environ. Sci. Ecotechnology, 2024, 22, 100443 71. J. Wang, D. Cheng, M. Gao, Q. Li, Y. Xin, N. Zhang, Z. Zhang, X. Yu, Z. Zhao, K. Zhou, Chem. Sci., 2021, 12, 3245 72. M. Nie, A. Cui, M. Wu, T. Guo, Q. Guo, J. CO2 Util., 2023, 75, 102579 73. C.-S. Chen, T.-C. Chen, J. H. Wu, H.-C. Wu, C.-M. Yang, T.-C. Yang, C.-W. Pao, Chem. Eng. J., 2024, 489, 151198 74. J. Zhang, S. Deo, M. J. Janik, J. W. Medlin, J. Am. Chem. Soc., 2020, 142, 5184 75. Z. Xie, S. Hwang, J. G. Chen, SmartMat., 2023, 4, e1201 76. Y. Zhang, Z. Zhang, X. Yang, R. Wang, H. Duan, Z. Shen, L. Li, Y. Su, R. Yang, Y. Zhang, X. Su, Y. Huang, T. Zhang, Green Chem., 2020, 22, 6855 77. W. Zhang, H. Lin, Y. Wei, X. Zhou, Y. An, Y. Dai, Q. Niu, T. Lin, L. Zhong, ACS Catal., 2024, 14, 2409 78. H. Xin, L. Lin, R. Li, D. Li, T. Song, R. Mu, Q. Fu, X. Bao, J. Am. Chem. Soc., 2022, 144, 4874 79. J. Zhao, X. Liu, Z. Li, K. Feng, Y. Pan, P. Ji, K. Zhao, B. Yan, D. Zhou, D. Su, ACS Catal., 2024, 14, 3158 80. Y. Shi, Z.-R. Ma, Y.-Y. Xiao, Y.-C. Yin, W.-M. Huang, Z.-C. Huang, Y.-Z. Zheng, F.-Y. Mu, R. Huang, G.-Y. Shi, Y.-Y. Sun, X.-H. Xia, W. Chen, Nat. Commun., 2021, 12, 3021 81. M. Shekhar, J. Wang, W.-S. Lee, W. D. Williams, S. M. Kim, E. A. Stach, J. T. Miller, W. N. Delgass, F. H. Ribeiro, J. Am. Chem. Soc., 2012, 134, 4700 82. L. Liu, A. Corma, Chem. Rev., 2018, 118, 4981 83. B. Yang, Y. Wang, B. Gao, L. Zhang, L. Guo, ACS Catal., 2023, 13, 10364 84. X. Y. Li, J. Lin, L. Li, Y. K. Huang, X. L. Pan, S. E. Collins, Y. J. Ren, Y. Su, L. L. Kang, X. Y. Liu, Y. L. Zhou, H. Wang, A. Q. Wang, B. T. Qiao, Xiaodong Wang, Tao Zhang, Angew. Chem. Int. Ed, 2020, 59, 19983 85. B. Han, Q. Li, X. Jiang, Y. Guo, Q. Jiang, Y. Su, L. Li, B. Qiao, Small, 2022, 18, 2204490 86. S. K. Liu, Q. Zhao, X. U. Han, C. Y. Wei, H. T. Liang, Y. D. Wang, S. Y. Huang, X. B. Ma, Trans. Tianjin Univ., 2023, 29, 293 87. C. Dong, R. Mu, R. Li, J. Wang, T. Song, Z. Qu, Q. Fu, X. Bao, J. Am. Chem. Soc., 2023, 145, 17056 88. S. X. Lin, Z. H. Li, M. S. Li, Fuel, 2023, 333, 126369 89. C. S. Yang, C. L. Pei, R. Luo, S. H. Liu, Y. N. Wang, Z. Y. Wang, Z. J. Zhao, J. L. Gong, J. Am. Chem. Soc., 2020, 142, 19523 90. J. Zhou, Z. Gao, G. L. Xiang, T. Y. Zhai, Z. K. Liu, W. X. Zhao, X. Liang, L. Y. Wang, Nat. Commun., 2022, 13, 327 91. Z. Y. Zhao, M. Z. Wang, P. J. Ma, Y. P. Zheng, J. Y. Chen, H. Q. Li, X. B. Zhang, K. Zheng, Q. Kuang, Z. X. Xie, Appl. Catal. , B, 2021, 291, 120101 92. Z. Han, C. Z. Tang, J. J. Wang, L. D. Li, C. Li, J. Catal., 2021, 394, 236-244 93. H. Zheng, W. Q. Liao, J. Q. Ding, F. K. Xu, A. P. Jia, W. X. Huang, Z. H. Zhang, ACS Catal, 2022, 12, 15451 94. W. Q. Liao, C. Tang, H. Zheng, J. Q. Ding, K. F. Zhang, H. W. Wang, J. Q. Lu, W. X. Huang, Z. H. Zhang, J. Catal., 2022, 407, 126 95. J. Zhang, T. F. Huang, R. Y. Wang, X. C. Li, J. Wang, H. Ge, J. Ren, Y. W. Wang, Z. F. Zheng, New J. Chem., 2023, 47, 11774 96. Y. J. Guo, L. Y. Jia, Z. Z. Zhang, M. Z. Gong, S. S. Dang, Y. Q. Huang, X. H. Gao, W. F. Tu, Y. F. Han, Chem. Eng. J., 2024, 491, 151964 97. S. N. Zhang, Z. X. Wu, X. F. Liu, Z. L. Shao, L. Xia, L. S. Zhong, H. Wang, Y. H. Sun, Appl. Catal. B, 2021, 293, 120207 98. G. Varvoutis, M. Lykaki, E. Papista, S. A. Carabineiro, A. C. Psarras, G. E. Marnellos, M. Konsolakis, J. CO2 Util., 2021, 44, 101408 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
Figure 1.
Possible reaction pathway for forming other molecules from carbon dioxide.
-
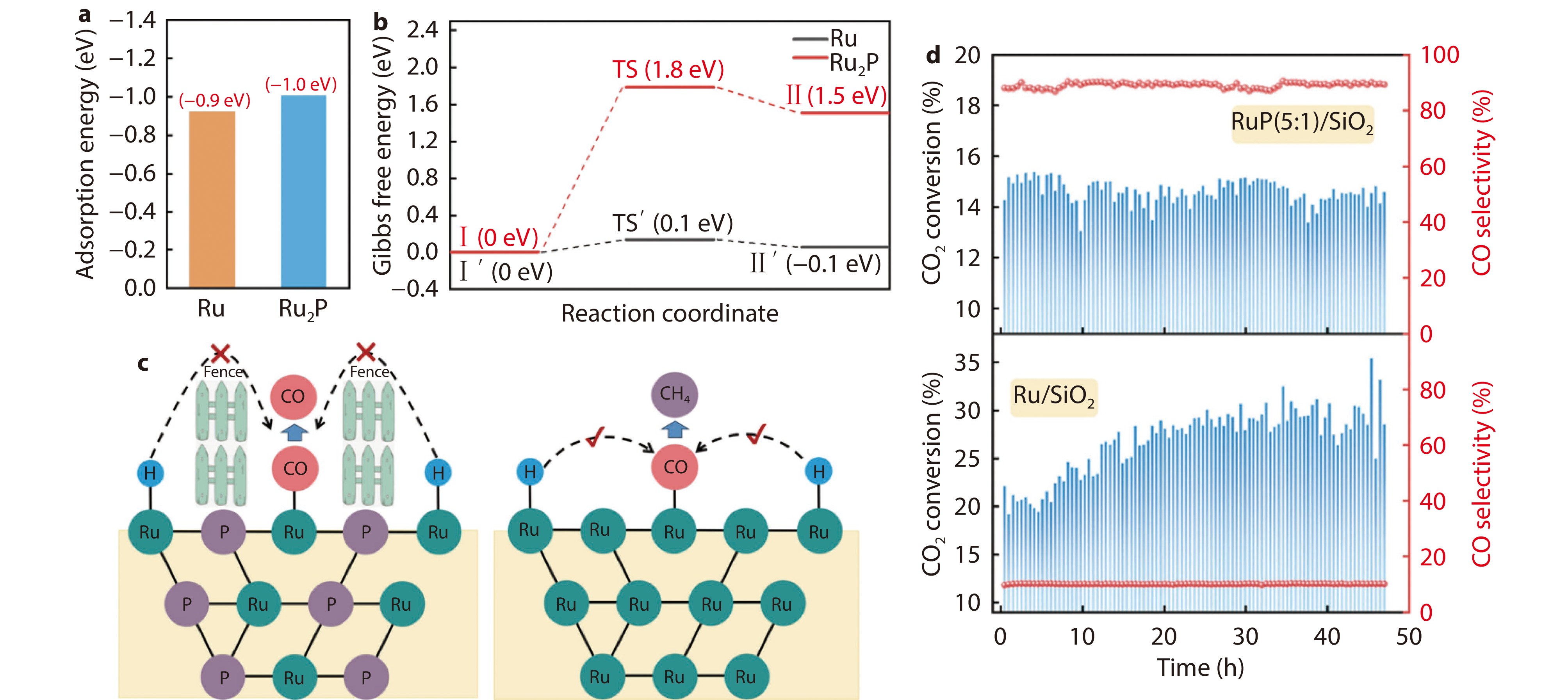
Figure 2.
a Calculation of H2 adsorption energy on Ru and Ru2P surfaces. b Gibbs free energy for migration of adsorbed H atoms. c Schematic diagram for the ‘fence effect’ of P on the migration of active H species and consequent changes in the CO2 hydrogenation selectivity. d Catalytic performance in long-term stability tests.[60] Copyright 2024, ACS Publications.
-
Figure 3.
Catalytic performances of the TiO2-supported a Ni-4.2 and b Ni-P-4.2 catalysts for CO2 hydrogenation as a function of temperature (1 vol % CO2 + 4 vol % H2 + 95 vol % N2 with a space velocity of ~30,000 mL gcat–1 h–1 at ambient pressure). c Comparison of activity and selectivity on various catalysts before and after secondary P modification (1 vol % CO2 + 4 vol % H2 + 95 vol % N2 at ambient pressure, 400 °C).[62] Copyright 2023, ACS Publications. Catalytic performances of Ru/TiO2-x catalysts for CO2 hydrogenation at d 200 °C, e 250 °C, and f 300 °C.[63] Copyright 2024, Springer Link.
-
Figure 4.
a Conversion rate, selectivity, and space-time yield (STY) with x% CoB/ZSM-5 catalysts and reference sample at different reaction temperatures. b Modification of Co nanoparticles with B atoms achieves selective reversal from CH4 to CO.[64] Copyright 2024, RSC Publishing. c The product selectivity on Ru/Ti-S catalyst with air and/or H2 pretreatment at 350 °C. d Schematic illustration of the mechanisms of CO2 hydrogenation on sulfate-free and sulfate-containing Ru/TiO2 catalysts.[65] Copyright 2024, Springer Nature.
-
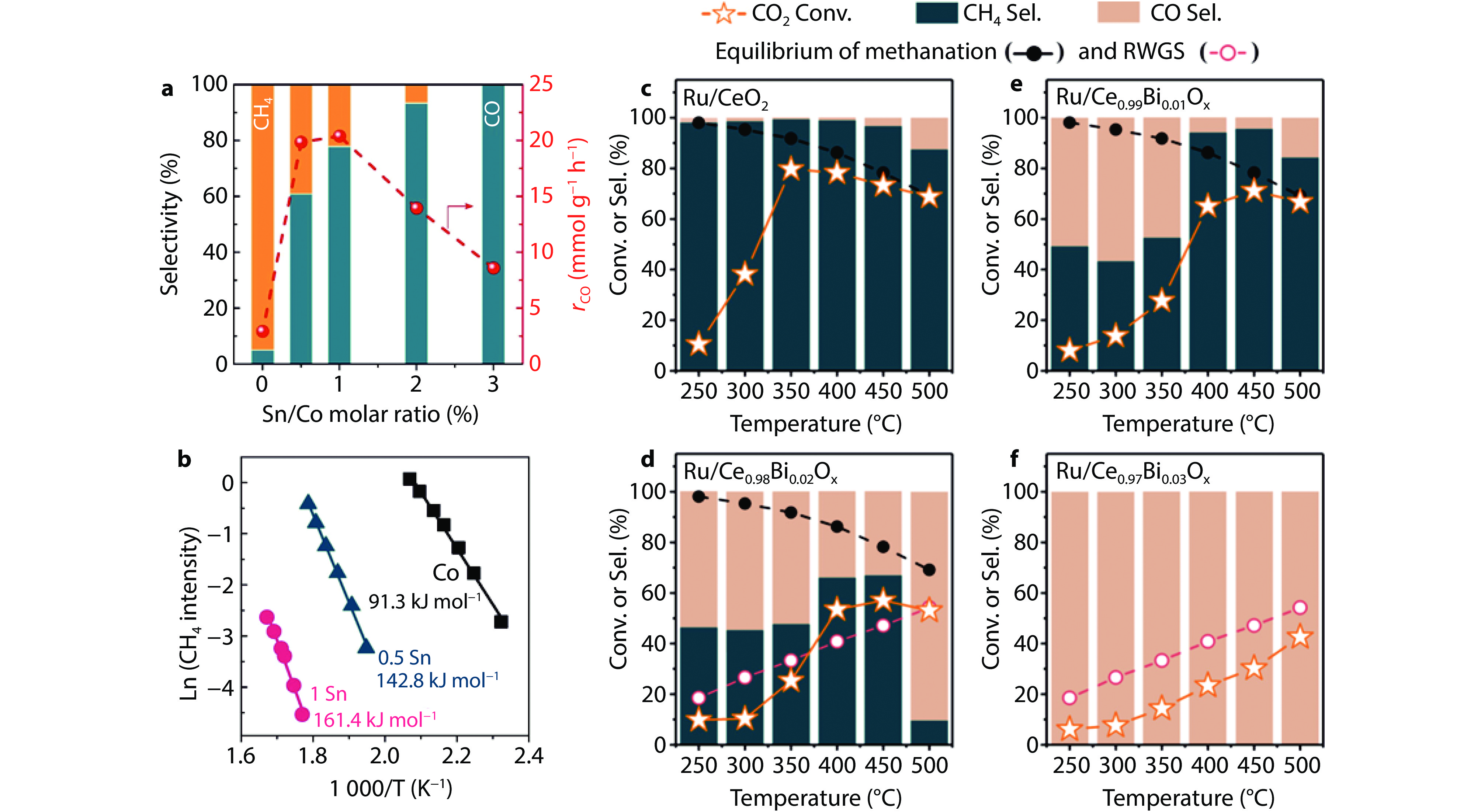
Figure 5.
a Effect of Sn addition on the product selectivity at ~15 % CO2 conversion level. Reaction conditions: T = 400 °C, P = 1 atm, GHSV was varied to obtain a similar CO2 conversion level of ~15 %. b Arrhenius plots for CH4 formation derived from CO2 + H2-TPSR at CO2 conversion <10 %.[66] Copyright 2023, Elsevier. CO2 hydrogenation performance evaluation over c Ru/CeO2, d Ru/Ce0.98Bi0.02Ox, e Ru/Ce0.99Bi0.01Ox, and f Ru/Ce0.97Bi0.03Ox. Reaction conditions: feed gas, CO2/H2 = 1/4; mcat = 150 mg, GHSV = 20,000 mL g–1 h–1.[67] Copyright 2024, ACS Publications.
-
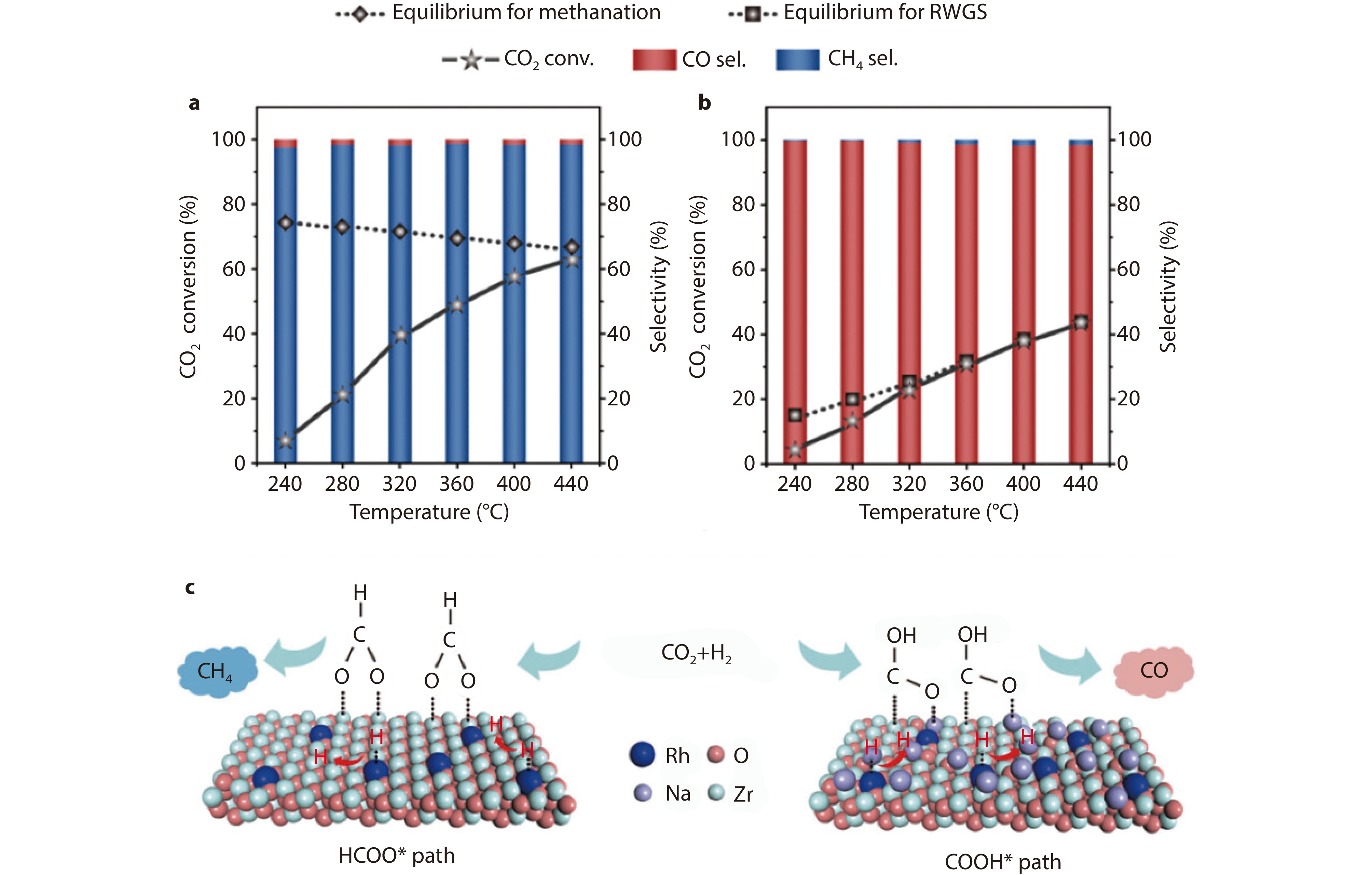
Figure 6.
The CO2 conversion and product selectivity of a Rh1/ZrO2 and b 1Na-Rh1/ZrO2 at different temperatures. Reaction conditions: catalyst, 100 mg; pressure, 1 MPa; feed gas, CO2/H2/Ar=24/72/4; WHSV, 18 000 mL gcat−1 h−1. Equilibrium conversions (dotted lines) as a function of temperature are plotted. c reaction mechanism. Na ions change the surface intermediate from HCOO* to COOH* and render the Rh atoms electron deficient with restrained H2 activation capability and weakened CO adsorption, thus cooperatively boosting CO formation.[68] Copyright 2022, Wiley.
-
Figure 7.
a Comparison of CO2 hydrogenation as a function of temperature on the Ni0.5SiO2 and Ni0.5Zrx/SiO2 catalysts in terms of the overall CO2 conversion.[73] Copyright 2024, Elsevier. Schemes showing: b CO selectivity with time on stream at different temperatures. black: 5%Pt/TiO2, red: NH2MPA/5%Pt/TiO2, yellow: BZPA/5%Pt/TiO2, blue: MPA/5%Pt/TiO2, catalyst loadings were kept at 80 mg for each case.[74] Copyright 2020, ACS Publications.
-
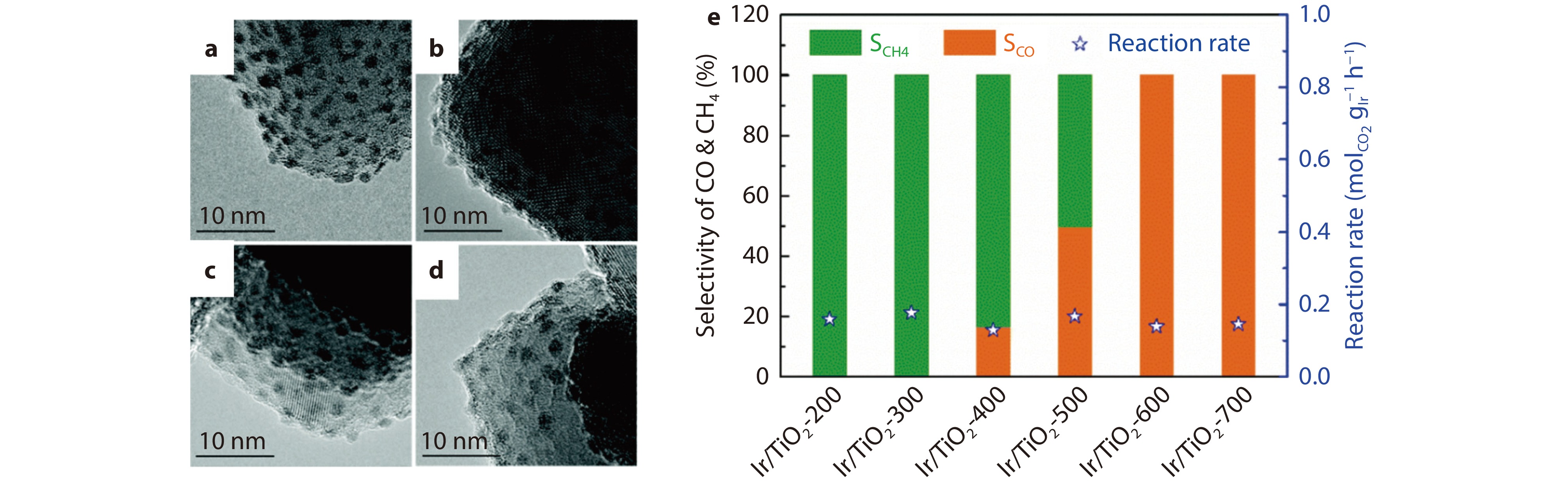
Figure 8.
HRTEM images of the series of Ir/TiO2-x catalysts: a Ir/TiO2-200, b Ir/TiO2-400, c Ir/TiO2-600, and d Ir/TiO2-700. e Catalytic performance of the series of Ir/TiO2-x catalysts for CO2 hydrogenation reactions. Reaction conditions: 280 °C, 0.1 MPa, space velocity =
9000 mL h−1 gcat−1, and H2/CO2/N2= 70/20/10.[76] Copyright 2020, RSC Publishing. -
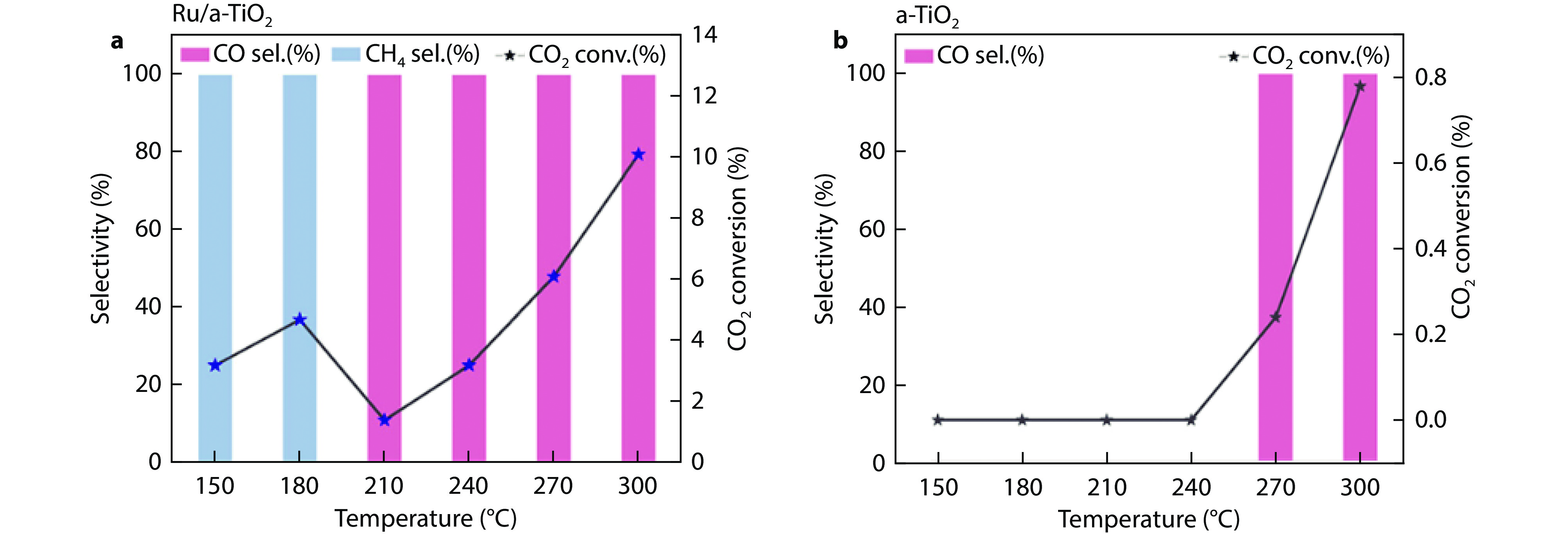
Figure 9.
Catalytic performance of various fresh Ru-based catalysts for CO2 hydrogenation. CO2 conversion and product selectivity of a Ru/a-TiO2 and b a-TiO2. Reaction conditions: 1.0 g, 1 MPa, and CO2/H2/Ar = 24/73/3,
3000 mL gcat–1 h–1.[77] Copyright 2024, ACS Publications. -
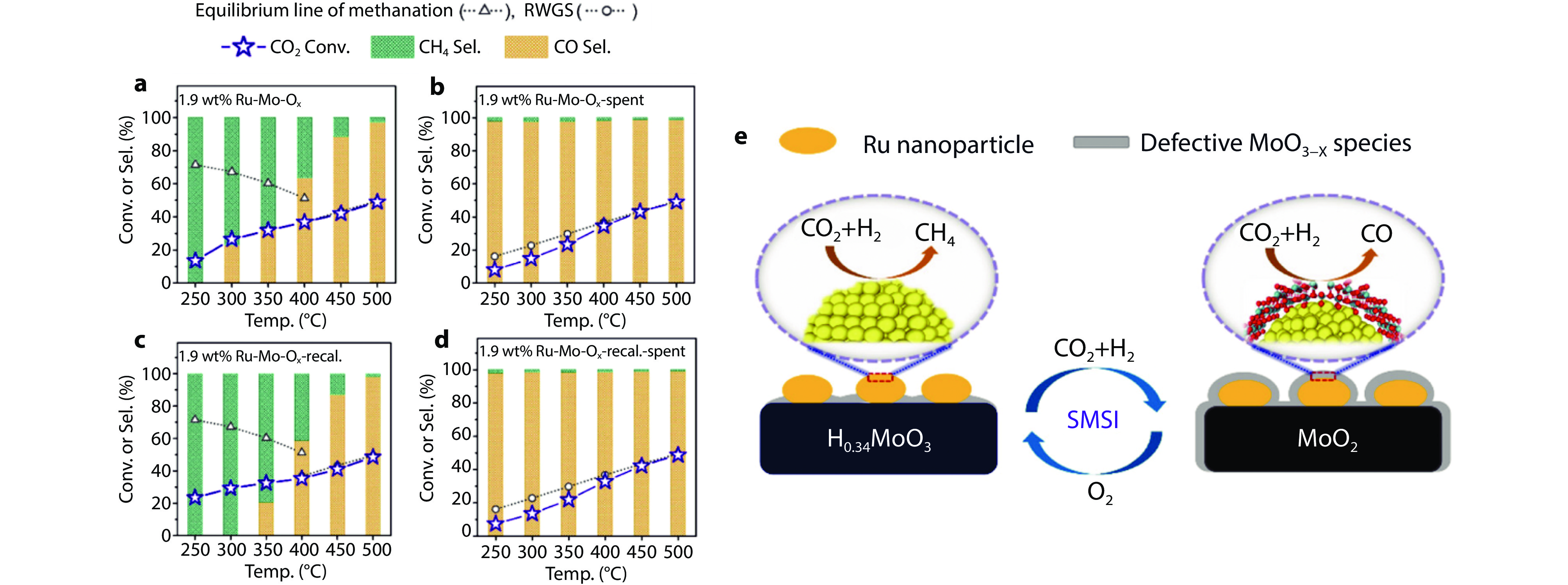
Figure 10.
Catalytic performance evolution of the Ru–Mo–oxide catalysts in CO2 hydrogenation. a Fresh 1.9 wt % Ru-Mo-Ox catalyst. b Spent catalyst after the reaction in a (1.9 wt %Ru-Mo-Ox-spent). c Spent catalyst calcined in O2 at 500 °C for 1 h (1.9 wt % Ru-Mo-Ox-recal). d Catalysts after the reaction in c (1.9 wt %Ru-Mo-Ox-recal.-spent). Reaction conditions: 50 mg of catalyst, 3% CO2/9% H2/N2, and WHSV = 10,000 mL gcatal-1 h-1. Catalytic performance of fresh 1.9 wt % Ru–Mo-Ox catalyst at 250 °C. e Selectivity changes in Ru/TiO2 catalysts.[78] Copyright 2022, ACS Publications.
-
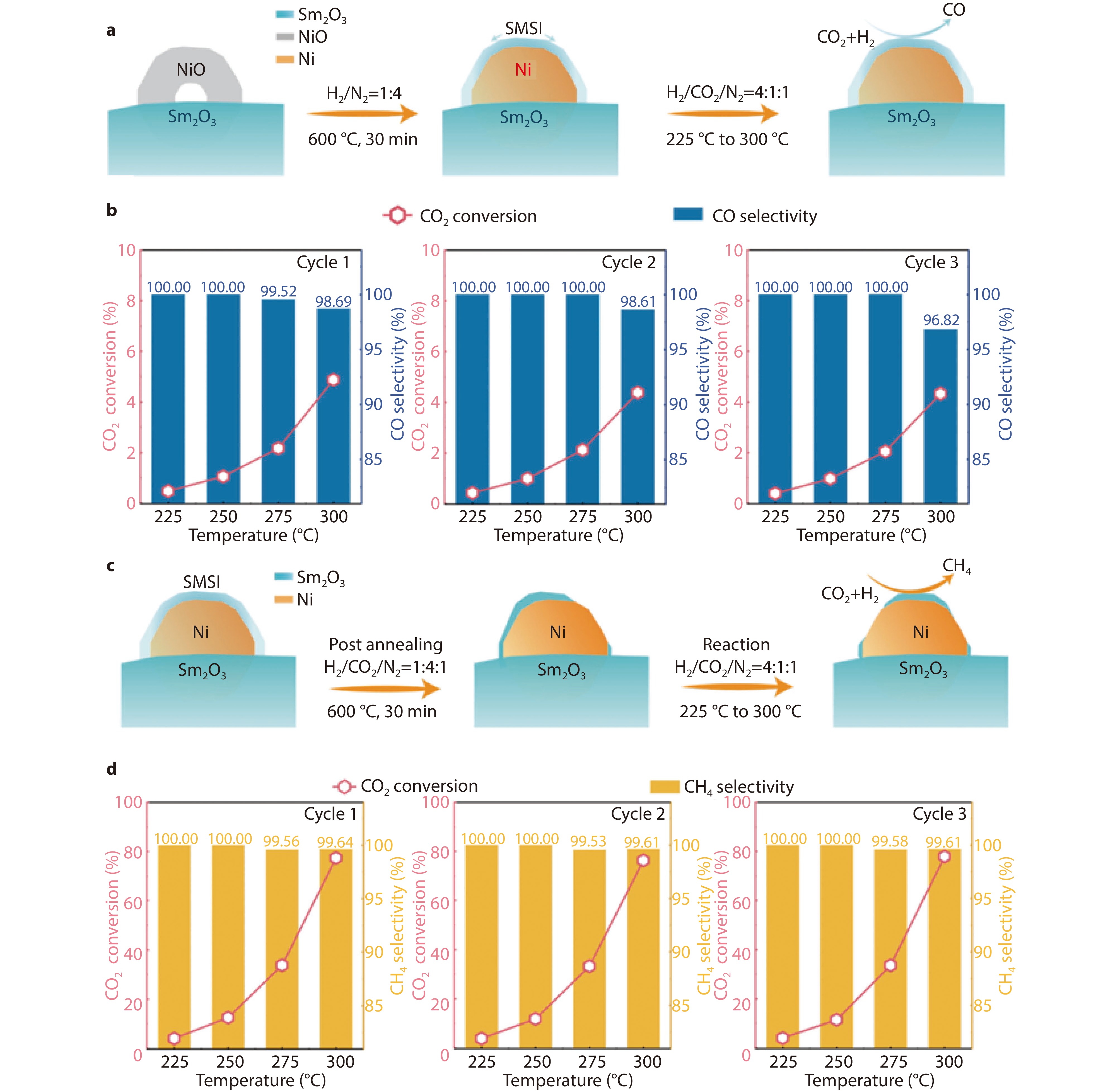
Figure 11.
a Schematic illustration of the Ni/Sm2O3 catalyst reduction process and the RWGS reaction. b Catalytic performance in CO2 hydrogenation under three start-up/shut–down cycles. Conversion rate and selectivity of the Ni/Sm2O3 catalyst. c Schematic illustration of the post-annealing process and CO2 methanation reaction over Ni/Sm2O3 catalyst. d Catalytic performance of CO2 methanation using Ni/Sm2O3 catalysts for three start-up/shut-down cycles.[79] Copyright 2024, ACS Publications.
-
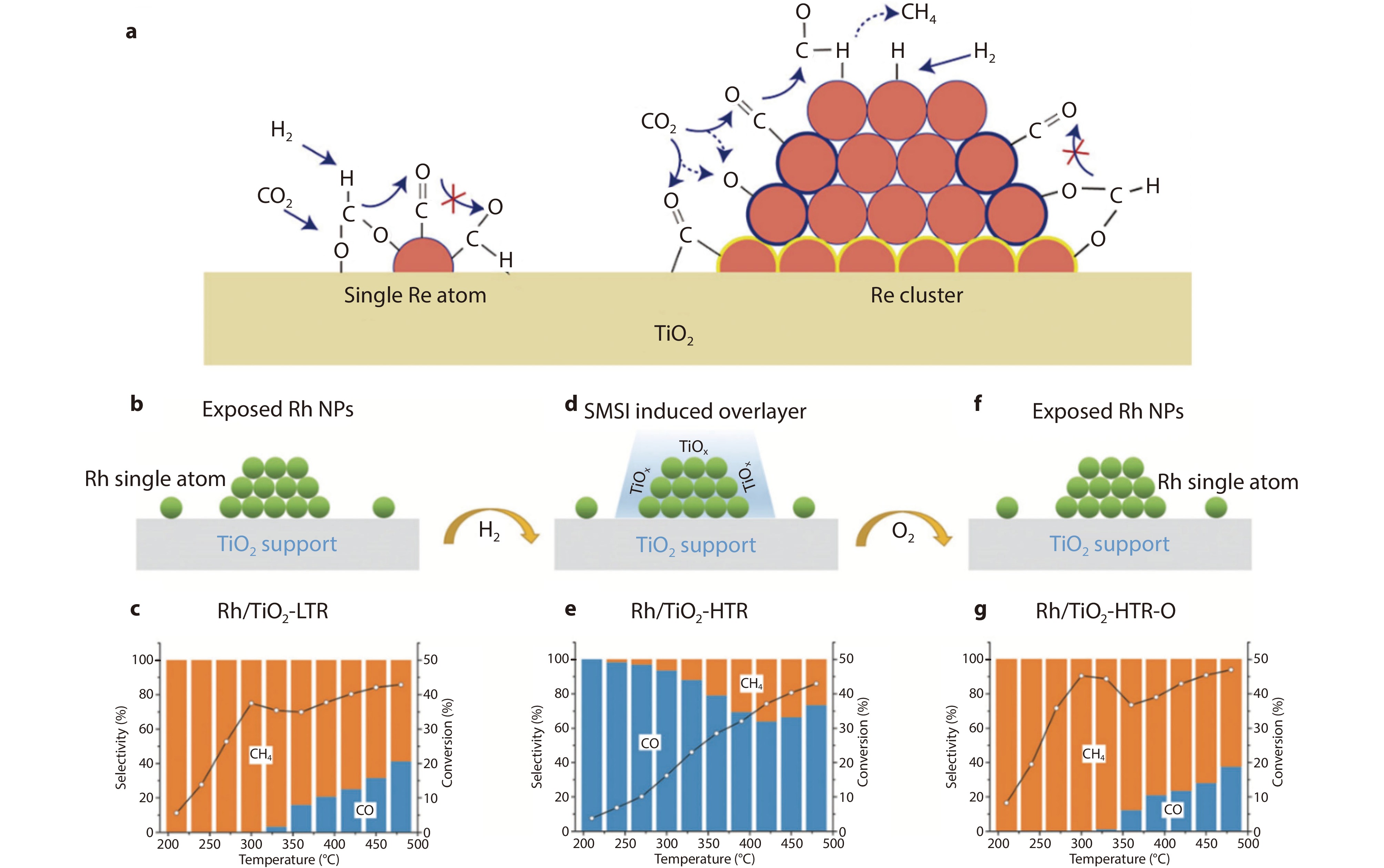
Figure 12.
a Influence of metal size on catalyst selectivity.[83] Copyright 2023, ACS Publications. b,d,f SMSI overlayer structures and catalytic performance of c Rh/TiO2-LTR, e Rh/TiO2-HTR and g Rh/TiO2-HTR-O in CO2 hydrogenation. Black open cycles represent CO2 conversion. Reaction conditions: P = 1 atm, 30 mg of catalyst, 4 vol% CO2 + 16 vol% H2 + 4 vol% N2 + He balance (N2 as internal standard, total flow rate = 30 mL min−1).[85] Copyright 2022, Wiley.
-
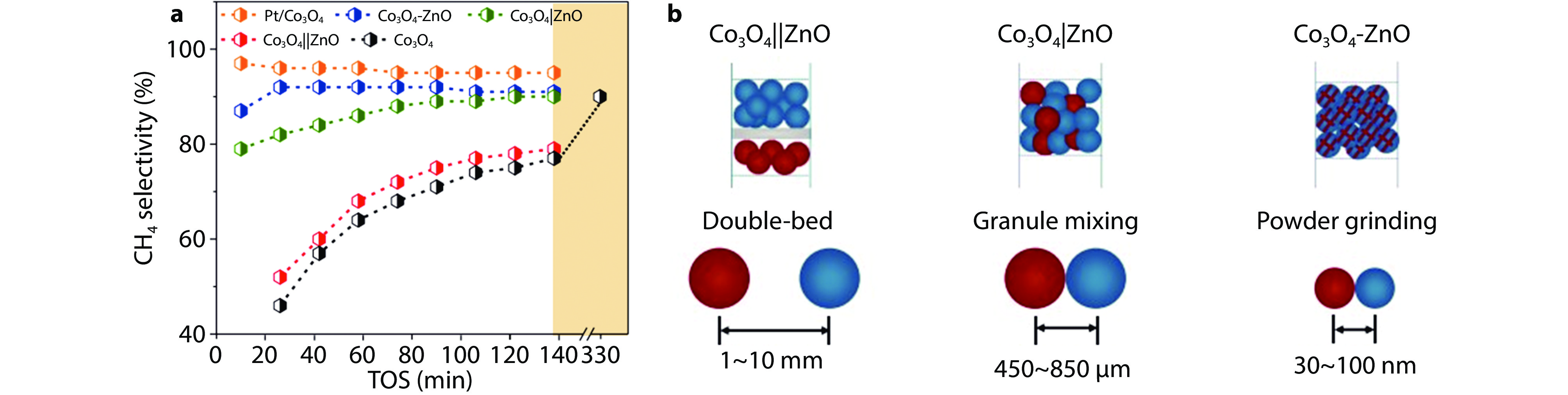
Figure 13.
a CH4 selectivity versus TOS of the Co3O4, Co3O4||ZnO, Co3O4|ZnO, Co3O4- ZnO and Pt/Co3O4 catalysts in CO2 hydrogenation. Reaction conditions: CO2/H2 = 1:3, P = 1 atm, T = 350 °C, WHSV = 32,000 mL gCo3O4-1 h-1. b Schematic display of the proximity.[87] Copyright 2023, ACS Publications.
-
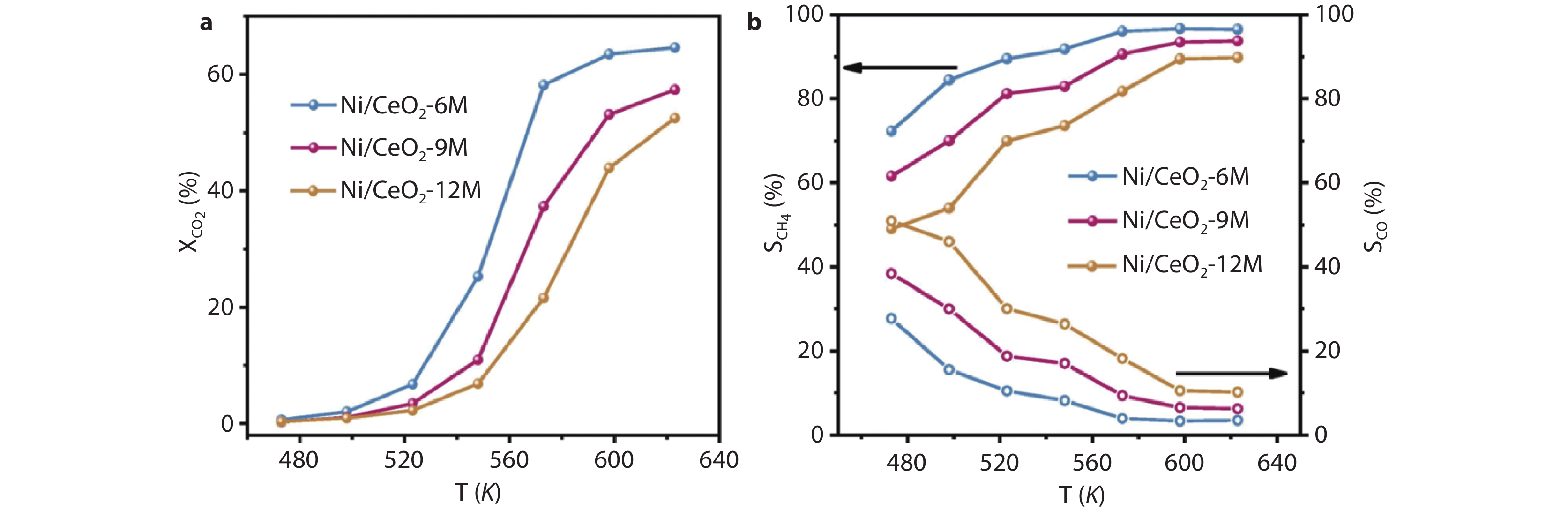
Figure 14.
Catalytic performance over Ni/CeO2 catalysts. a CO2 conversion and b CH4/CO selectivity as a function of reaction temperature (T = 473-623 K) Reaction conditions: T reduced = 723 K, P = 0.1 MPa, WHSV = 36 L g-1 h-1.[88] Copyright 2023, Elsevier.
-
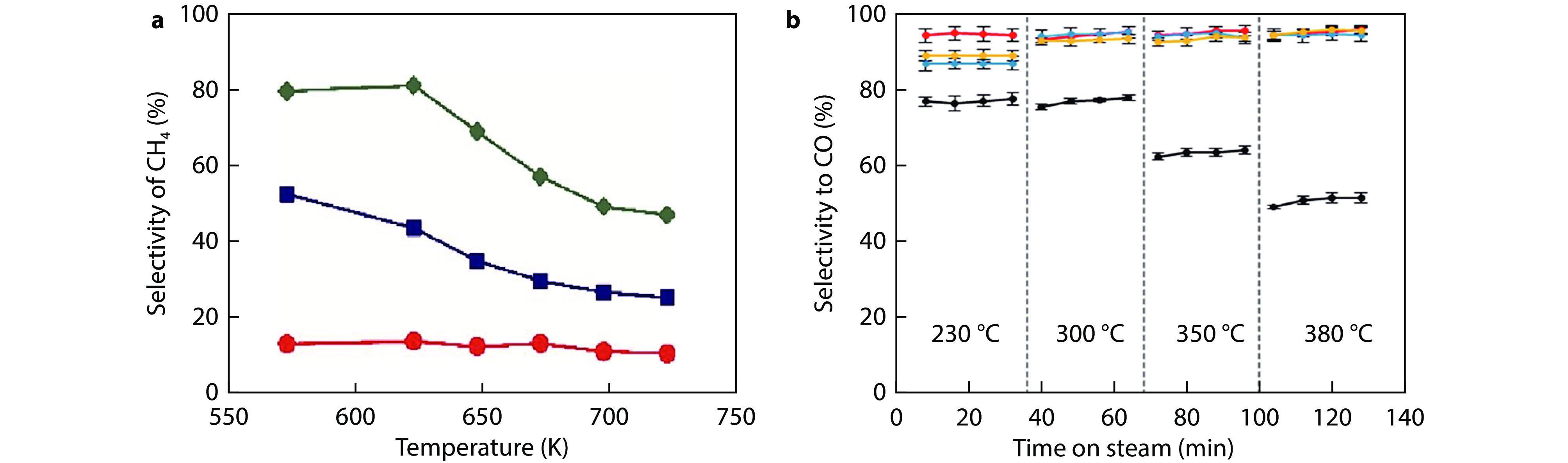
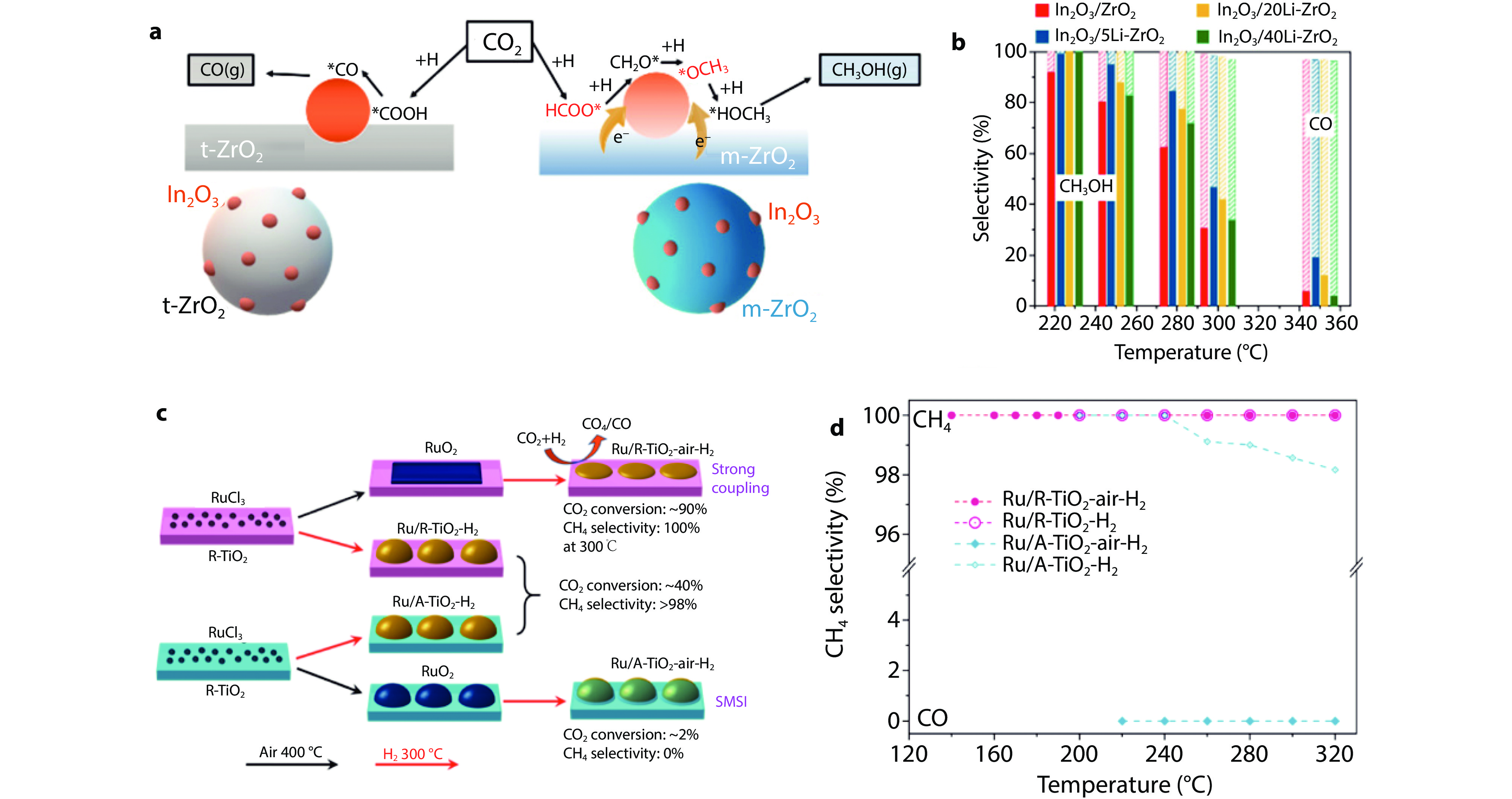
Figure 15.
a Simplified models showing the main reaction mechanism on In2O3/t-ZrO2 and In2O3/m-ZrO2. b Product distribution for CO2 hydrogenation over different catalysts. Methanol selectivity: solid bar, CO selectivity: shaded bar.[89] Copyright 2020, ACS Publications. c Temperature-dependent CH4 selectivity. d Summary scheme of varied activity and selectivity by direct H2 reduction and after annealing in air. Ru/TiO2–H2 refers to directly reduced catalysts by H2, while Ru/TiO2–air–H2 refers to catalysts by annealing in air at 400 °C and further reduction by H2.[90] Copyright 2022, Springer Nature.
-
Figure 16.
a-b Temperature programmed RWGS reaction over Pt/CeO2 catalysts.[91] Copyright 2021, Elsevier. c Methanol and CO selectivity of the catalysts. All the catalysts were tested under conditions of T = 300 °C, P = 2 MPa, and SV = 24,000 mL h−1 gcat−1.[92] Copyright 2021, Elsevier. d Catalytic activity and product selectivity of CO2 hydrogenation over various Ni/CeO2 catalysts at 290 °C.[93] Copyright 2022, ACS Publications.
-
Figure 17.
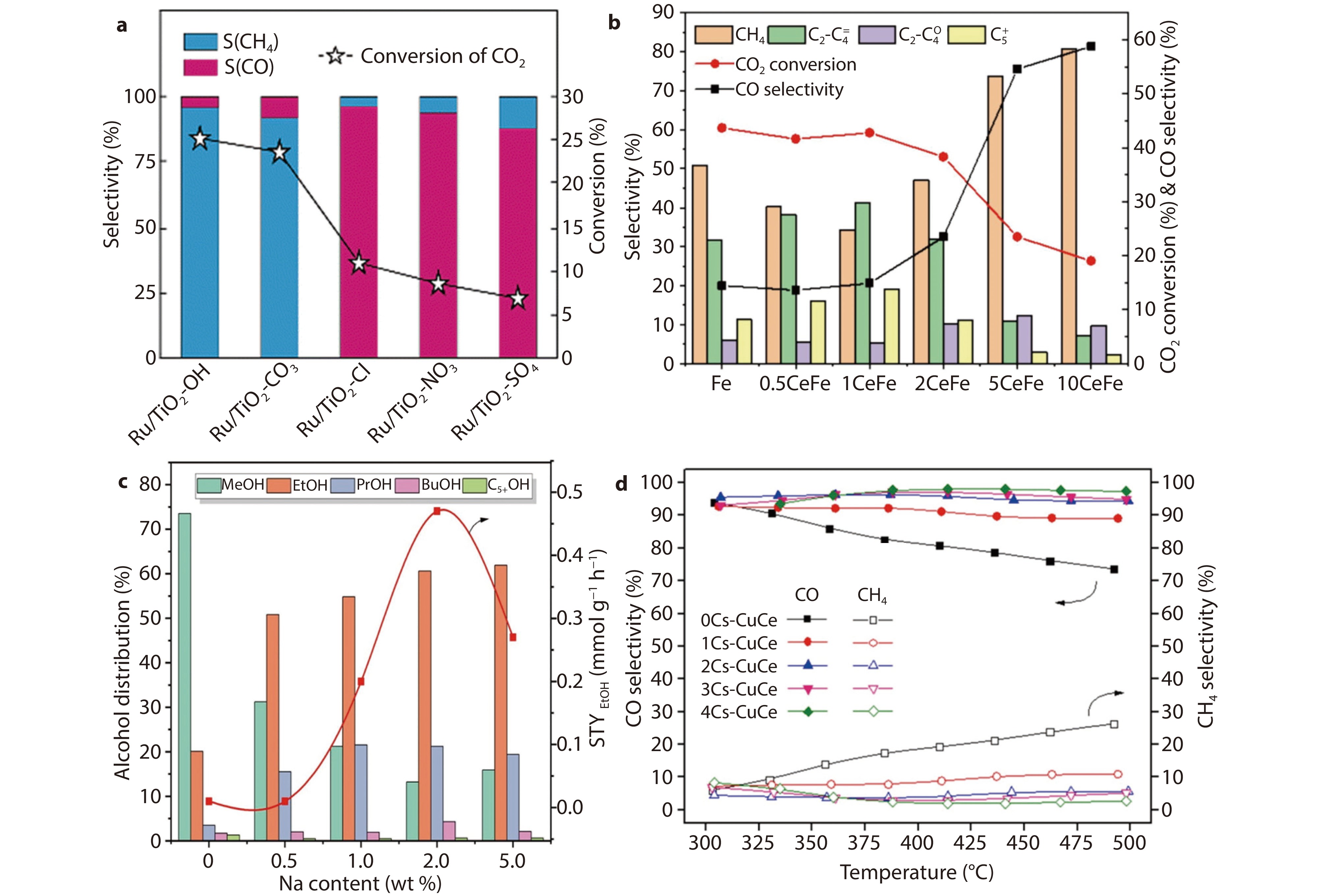
a Catalytic performances of Ru/TiO2 catalysts modified by various anions for CO2 hydrogenation at 300 °C.[95] Copyright 2023, RSC Publishing. b The catalytic activities of catalysts with different Ce contents.[96] Copyright 2024, Elsevier. c Alcohol distribution and ethanol STY. Catalytic performance under the conditions of 250 °C, 5 MPa, WHSV =
6000 mL g−1 h−1, and H2/CO2/N2 = 73/24/3.[97] Copyright 2021, Elsevier. d Effect of temperature on CO2 product distribution for the as-prepared samples. Reaction conditions: H2:CO2 = 9:1, WHSV = 30 L g–1 h−1, P =1 atm.[98] Copyright 2021, Elsevier.

 Xiyu Wang receibed his BSc degree from Liaoning University in 2024. He is currently pursuing his master's degree at the School of Applied Chemistry and Engineering, University of Science and Technology of China (Changchun Institute of Applied Chemistry, Chinese Academy of Sciences (CAS)) under the supervision of Prof. Xiao Wang. His current research interests focus on the rare earth-based catalytic nanomaterials for heterogeneous catalytic reactions.
Xiyu Wang receibed his BSc degree from Liaoning University in 2024. He is currently pursuing his master's degree at the School of Applied Chemistry and Engineering, University of Science and Technology of China (Changchun Institute of Applied Chemistry, Chinese Academy of Sciences (CAS)) under the supervision of Prof. Xiao Wang. His current research interests focus on the rare earth-based catalytic nanomaterials for heterogeneous catalytic reactions.  Lingling Zhang received her BSc and PhD from Jilin University in 2017 and 2022 respectively. She is currently working under the direction of Prof. Xiao Wang at Changchun Institute of Applied Chemistry, CAS. Her research interests focus on the preparation of rare earth-based heterogeneous catalysts.
Lingling Zhang received her BSc and PhD from Jilin University in 2017 and 2022 respectively. She is currently working under the direction of Prof. Xiao Wang at Changchun Institute of Applied Chemistry, CAS. Her research interests focus on the preparation of rare earth-based heterogeneous catalysts.  Shuyan Song received his BSc degree in Chemistry in 2003 and an MSc degree in Inorganic Chemistry in 2006, both from Northeast Normal University. He joined the group of Prof. Hongjie Zhang at Changchun Institute of Applied Chemistry, Chinese Academy of Sciences (CAS), where he received his Ph.D. degree in Inorganic Chemistry in 2009. He is working as a full professor at Changchun Institute of Applied Chemistry, CAS. His research focus is primarily on the development of porous functional materials for heterogeneous catalysis, proton conduction, chemical sensing and detection.
Shuyan Song received his BSc degree in Chemistry in 2003 and an MSc degree in Inorganic Chemistry in 2006, both from Northeast Normal University. He joined the group of Prof. Hongjie Zhang at Changchun Institute of Applied Chemistry, Chinese Academy of Sciences (CAS), where he received his Ph.D. degree in Inorganic Chemistry in 2009. He is working as a full professor at Changchun Institute of Applied Chemistry, CAS. His research focus is primarily on the development of porous functional materials for heterogeneous catalysis, proton conduction, chemical sensing and detection. 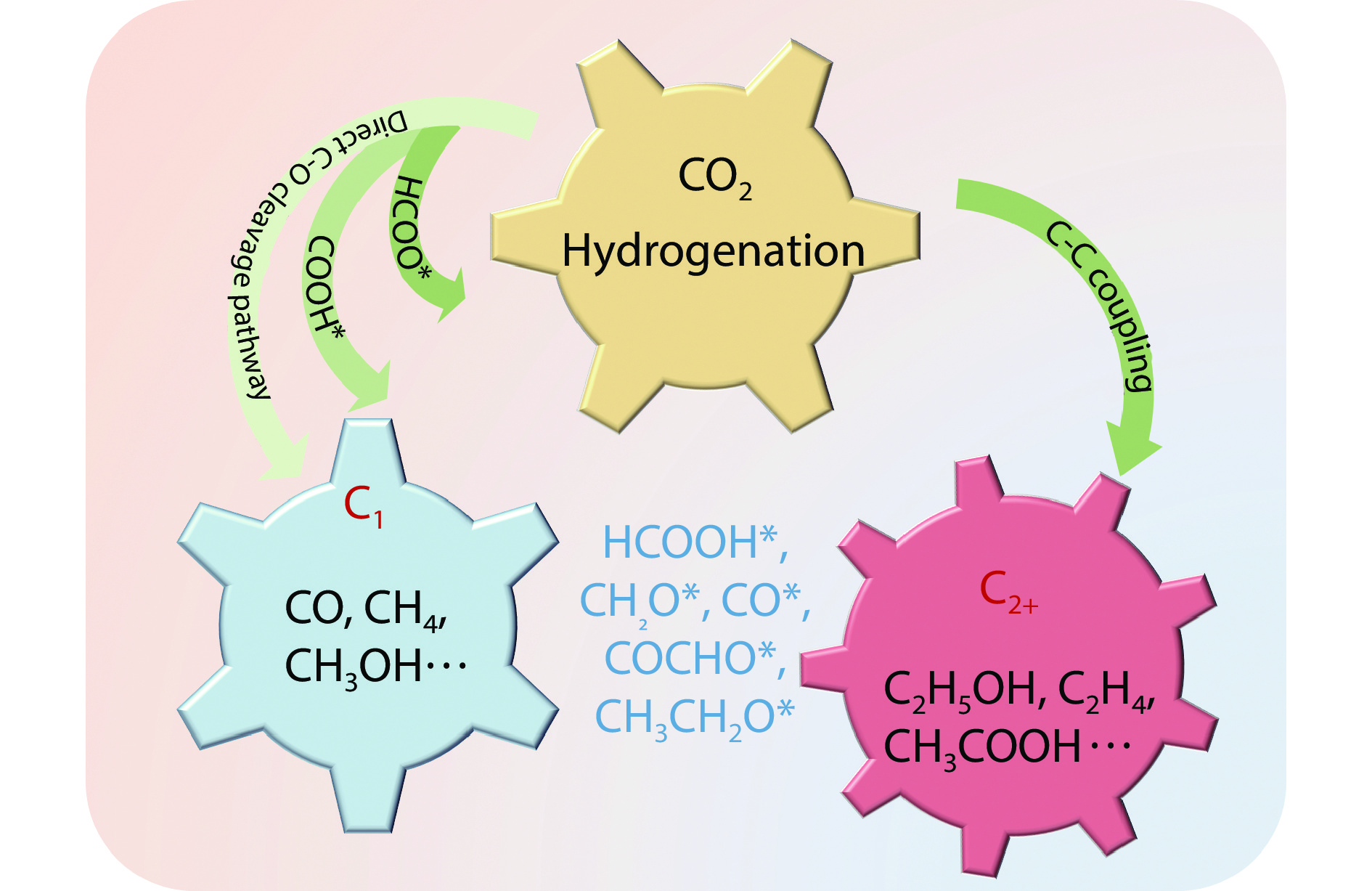

 DownLoad:
DownLoad: