| Citation: | Guodong Zhu, Qinghua Zhang, Lin Gu. Electron microscopy measurements of electron orbitals[J]. Materials Lab, 2025, 4(3): 250002. doi: 10.54227/mlab.20250002 |
Electron microscopy measurements of electron orbitals
-
Abstract
Quantitative characterization and real-space mapping of the electron orbital are highly desirable as it’s a basic order parameter linking microscopic electronic structure and macroscopic properties in material science. Here we briefly review the recent progress in the measurements of orbital populations via electron microscopic methods, including quantitative convergent-beam electron diffraction (QCBED), electron energy-loss spectroscopy (EELS) and others. Combing with the rapid development of four-dimensional scanning transmission electron microscopy (4D-STEM), we propose the future potential directions and discuss possible solutions towards real-space orbital mapping at atomic scale.
-
-
References
1. D. Xiao, L. Gu, Nano Sel., 2020, 1, 183 2. Y. Tokura, M. Kawasaki, N. Nagaosa, Nat. Phys., 2017, 13, 1056 3. Z. Chen, Y. Jiang, Y. T. Shao, M. E. Holtz, M. Odstrcil, M. Guizar-Sicairos, I. Hanke, S. Ganschow, D. G. Schlom, D. A. Muller, Science, 2021, 372, 826 4. W. F. Yang, H. Z. Sha, J. Z. Cui, L. Z. Mao, R. Yu, Nat. Nanotechnol., 2024, 19, 612 5. E. Clementi, G. Corongiu, Int. J. Quantum Chem., 2008, 108, 1758 6. J. M. Zuo, M. Kim, M. O'Keeffe, J. C. H. Spence, Nature, 1999, 401, 49 7. C. J. Humphreys, Nature, 1999, 401, 21 8. J. M. Zuo, M. O'Keeffe, M. Kim, J. C. H. Spence, Angew. Chem. Int. Ed., 2000, 39, 3791 9. W. H. E. Schwarz, Angew. Chem. Int. Ed., 2006, 45, 1508 10. M. Labarca, O. Lombardi, Found. Chem., 2010, 12, 149 11. C. E. Brion, G. Cooper, Y. Zheng, I. V. Litvinyuk, I. E. McCarthy, Chem. Phys., 2001, 270, 13 12. J. Itatani, J. Levesque, D. Zeidler, H. Niikura, H. Pépin, J. C. Kieffer, P. B. Corkum, D. M. Villeneuve, Nature, 2004, 432, 867 13. T. Manjo, S. Kitou, N. Katayama, S. Nakamura, T. Katsufuji, Y. Nii, T. -H. Arima, J. Nasu, T. Hasegawa, K. Sugimoto, D. Ishikawa, A. Q. R. Baron, H. Sawa, Mater. Adv., 2022, 3, 3192 14. M. Bugnet, S. Löffler, M. Ederer, D. M. Kepaptsoglou, Q. M. Ramasse, J. Microsc., 2024, 295, 217 15. Y. Tokura, N. Nagaosa, Science, 2000, 288, 462 16. J. Zhu, Y. Miao, J. T. Guo, Acta Mater., 1997, 45, 1989 17. J. M. Zuo, Mater. Trans. Jim, 1998, 39, 938 18. F. Feng, A. H. Zhang, J. Zhu, Ultramicroscopy, 2004, 98, 173 19. J. M. Zuo, Rep. Prog. Phys., 2004, 67, 2053 20. L. J. Wu, Y. M. Zhu, J. Tafto, Phys. Rev. B, 1999, 59, 6035 21. Y. M. Zhu, L. J. Wu, J. Tafto, Microsc. Microanal., 2003, 9, 442 22. L. J. Wu, Y. M. Zhu, T. Vogt, H. B. Su, J. W. Davenport, J. Tafto, Phys. Rev. B, 2004, 69, 064501 23. K. Tsuda, Y. Ogata, K. Takagi, T. Hashimoto, M. Tanaka, Acta Cryst. A, 2002, 58, 514 24. F. Yu, H. M. Zou, J. B. Wang, R. H. Wang, Micron, 2004, 35, 411 25. F. Feng, J. Zhu, A. H. Zhang, Acta Cryst. A, 2005, 61, 453 26. J. F. Cao, H. M. Zou, C. Guo, Z. L. Chen, S. Z. Pu, Solid State Ionics, 2009, 180, 1209 27. P. N. H. Nakashima, A. E. Smith, J. Etheridge, B. C. Muddle, Science, 2011, 331, 1583 28. X. Sang, A. Kulovits, G. Wang, J. Wiezorek, Philos. Mag., 2012, 92, 4408 29. D. Peng, P. N. H. Nakashima, Phys. Rev. Lett., 2021, 126, 176402 30. N. K. Hansen, P. Coppens, Acta Cryst. A, 1978, 34, 909 31. A. Holladay, P. Leung, P. Coppens, Acta Cryst. A, 1983, 39, 377 32. T. Shang, D. Xiao, F. Meng, X. Rong, A. Gao, T. Lin, Z. Tang, X. Liu, X. Li, Q. Zhang, Y. Wen, R. Xiao, X. Wang, D. Su, Y.-S. Hu, H. Li, Q. Yu, Z. Zhang, V. Petricek, L. Wu, L. Gu, J.-M. Zuo, Y. Zhu, C.-W. Nan, J. Zhu, Nat. Commun., 2022, 13, 5810 33. Z. Tang, T. Shang, H. Xu, T. Lin, A. Gao, W. Lin, X. Li, S. Wang, B. Yu, F. Meng, Q. Zhang, X. Wang, D. Su, Q. Meng, L. Wu, L. Gu, C. W. Nan, Angew. Chem. Int. Ed., 2023, 62, e202216898 34. T. Shang, A. Gao, D. Xiao, Q. Zhang, X. Rong, Z. Tang, W. Lin, T. Lin, F. Meng, X. Li, Y. Wen, X. Wang, D. Su, Z. Chen, Y.-S. Hu, H. Li, Q. Yu, Z. Zhang, L. Wu, L. Gu, J.-M. Zuo, Y. Zhu, L. Chen, C.-W. Nan, Natl. Sci. Rev., 2024, 11, nwae255 35. R. F. Egerton, Ultramicroscopy, 2012, 119, 24 36. J. C. H. Spence, P. W. Hawkes, Ultramicroscopy, 2008, 108, 1502 37. S. Löffler, V. Motsch, P. Schattschneider, Ultramicroscopy, 2013, 131, 39 38. L. Pardini, S. Löffler, G. Biddau, R. Hambach, U. Kaiser, C. Draxl, P. Schattschneider, Phys. Rev. Lett., 2016, 117, 036801 39. S. Löffler, M. Bugnet, N. Gauquelin, S. Lazar, E. Assmann, K. Held, G. A. Botton, P. Schattschneider, Ultramicroscopy, 2017, 177, 26 40. J. Fink, Adv. Electron. Electron Phys., 1989, 75, 121 41. J. Fink, N. Nucker, E. Pellegrin, H. Romberg, M. Alexander, M. Knupfer, J. Electron. Spectrosc., 1994, 66, 395 42. A. Torres-Pardo, A. Gloter, P. Zubko, N. Jecklin, C. Lichtensteiger, C. Colliex, J. M. Triscone, O. Stéphan, Phys. Rev. B, 2011, 84, 220102 43. K. Xu, L. Zhang, A. Godfrey, D. Song, W. Si, Y. Zhao, Y. Liu, Y. Rao, H. Zhang, H. A. Zhou, W. Jiang, W. Wang, Z. Cheng, J. Zhu, Proc. Natl. Acad. Sci. U.S.A., 2021, 118, e2101106118 44. C. Iwashimizu, M. Haruta, H. Kurata, Appl. Phys. Lett., 2021, 119, 232902 45. M. Bugnet, M. Ederer, V. K. Lazarov, L. Li, Q. M. Ramasse, S. Löffler, D. M. Kepaptsoglou, Phys. Rev. Lett., 2022, 128, 116401 46. M. Ederer, S. Löffler, Ultramicroscopy, 2024, 256, 113866 47. A. J. D'Alfonso, B. Freitag, D. Klenov, L. J. Allen, Phys. Rev. B, 2010, 81, 100101 48. J. S. Jeong, M. L. Odlyzko, P. Xu, B. Jalan, K. A. Mkhoyan, Phys. Rev. B, 2016, 93, 165140 49. H. J. Lee, W. Ho, Science, 1999, 286, 1719 50. J. Repp, G. Meyer, S. Paavilainen, F. E. Olsson, M. Persson, Science, 2006, 312, 1196 51. L. Gross, N. Moll, F. Mohn, A. Curioni, G. Meyer, F. Hanke, M. Persson, Phys. Rev. Lett., 2011, 107, 086101 52. H. Kim, Y. Yoshida, C. C. Lee, T. R. Chang, H. T. Jeng, H. Lin, Y. Haga, Z. Fisk, Y. Hasegawa, Sci. Adv., 2017, 3, eaao0362 53. S. Wang, N. Kennedy, K. Fujita, S.-I. Uchida, H. Eisaki, P. D. Johnson, J. C. S. Davis, S. M. O’Mahony, Nat. Mater., 2024, 23, 492 54. H. Hafiz, K. Suzuki, B. Barbiellini, Y. Orikasa, V. Callewaert, S. Kaprzyk, M. Itou, K. Yamamoto, R. Yamada, Y. Uchimoto, Y. Sakurai, H. Sakurai, A. Bansil, Sci. Adv., 2017, 3, e1700971 55. H. Yavaş, M. Sundermann, K. Chen, A. Amorese, A. Severing, H. Gretarsson, M. W. Haverkort, L. H. Tjeng, Nat. Phys., 2019, 15, 559 56. S. Kitou, T. Manjo, N. Katayama, T. Shishidou, T.-H. Arima, Y. Taguchi, Y. Tokura, T. Nakamura, T. Yokoyama, K. Sugimoto, H. Sawa, Phys. Rev. Res., 2020, 2, 033503 57. S. Kitou, Y. Kaneko, Y. Nakamura, K. Sugimoto, Y. Nomura, R. Arita, Y. Tokura, H. Sawa, T. H. Arima, Phys. Rev. B, 2023, 108, 024103 58. P. Coppens, B. Iversen, F. K. Larsen, Coordin. Chem. Rev., 2005, 249, 179 59. R. Kaminski, S. Domagala, K. N. Jarzembska, A. A. Hoser, W. F. Sanjuan-Szklarz, M. J. Gutmann, A. Makal, M. Malinska, J. M. Bak, K. Wozniak, Acta Cryst. A, 2014, 70, 72 60. H. Kasai, K. Tolborg, M. Sist, J. Zhang, V. R. Hathwar, M. Ø. Filsø, S. Cenedese, K. Sugimoto, J. Overgaard, E. Nishibori, B. B. Iversen, Nat. Mater., 2018, 17, 249 61. C. Gao, A. Genoni, S. Gao, S. Jiang, A. Soncini, J. Overgaard, Nat. Chem., 2020, 12, 213 62. C. Ophus, Microsc. Microanal., 2019, 25, 563 63. K. M. Müller-Caspary, M. Duchamp, M. Rösner, V. Migunov, F. Winkler, H. Yang, M. Huth, R. Ritz, M. Simson, S. Ihle, H. Soltau, T. Wehling, R. E. D. Dunin-Borkowski, S. Van Aert, A. Rosenauer, Phys. Rev. B, 2018, 98, 121408 64. S. Fang, Y. Wen, C. S. Allen, C. Ophus, G. G. D. Han, A. I. Kirkland, E. Kaxiras, J. H. Warner, Nat. Commun., 2019, 10, 1127 65. W. P. Gao, C. Addiego, H. Wang, X. X. Yan, Y. S. Hou, D. X. Ji, C. Heikes, Y. Zhang, L. Z. Li, H. X. Huyan, T. Blum, T. Aoki, Y. F. Nie, D. G. Schlom, R. Q. Wu, X. Q. Pan, Nature, 2019, 575, 480 66. J. Martis, S. Susarla, A. Rayabharam, C. Su, T. Paule, P. Pelz, C. Huff, X. T. Xu, H. K. Li, M. Jaikissoon, V. Chen, E. Pop, K. Saraswat, A. Zettl, N. R. Aluru, R. Ramesh, P. Ercius, A. Majumdar, Nat. Commun., 2023, 14, 4363 67. L. Susana, A. Gloter, M. Tencé, A. Zobelli, ACS Nano, 2024, 18, 7424 68. S. Löffler, M. Ederer, Microsc. Microanal., 2023, 29, 376 69. L. J. Wu, Q. P. Meng, Y. M. Zhu, Ultramicroscopy, 2020, 219, 113095 70. T. Friedrich, C.-P. Yu, J. Verbeeck, S. Van Aert, Microsc. Microanal., 2023, 29, 395 71. B. Diederichs, Z. Herdegen, A. Strauch, F. Filbir, K. Müller-Caspary, Nat. Commun., 2024, 15, 101 -
Rights and permissions
This is an open access article under the terms of the Creative Commons Attribution License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited.
Information
Article Metrics
-
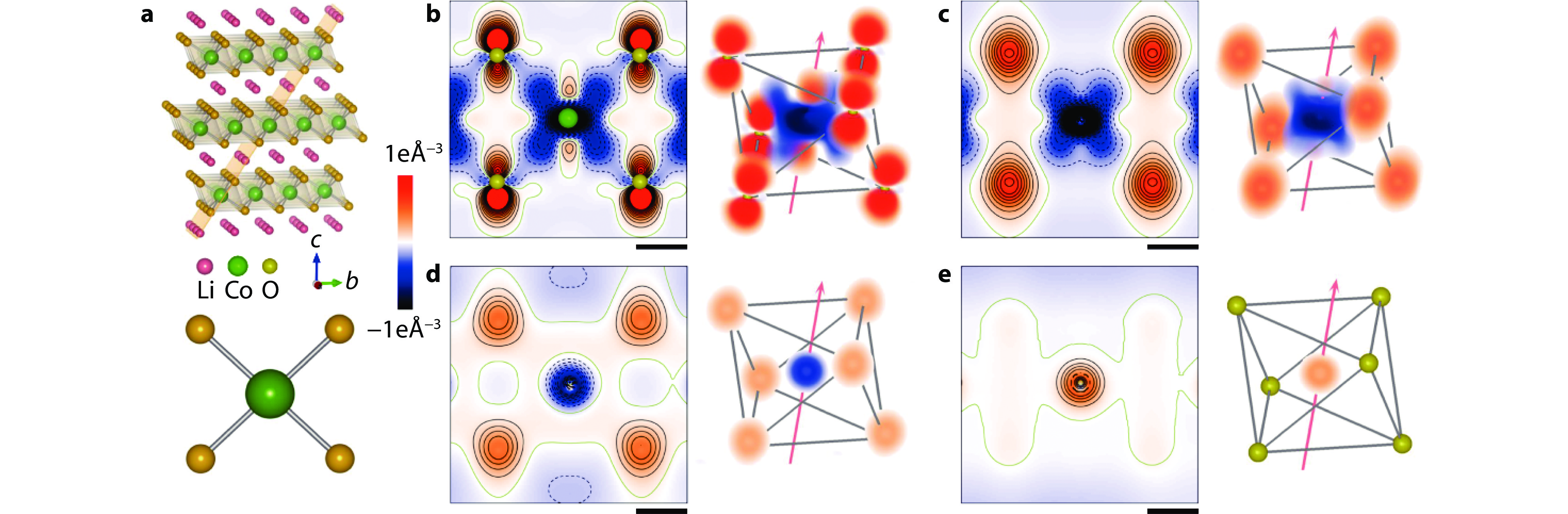
Figure 1.
Electron density evolution of the Co–O and O–O interactions. a Structural model of LiCoO2 with the indexed (
$ 01\stackrel{-}{4} $ $ 01\stackrel{-}{4} $ $ 01\stackrel{-}{4} $ -
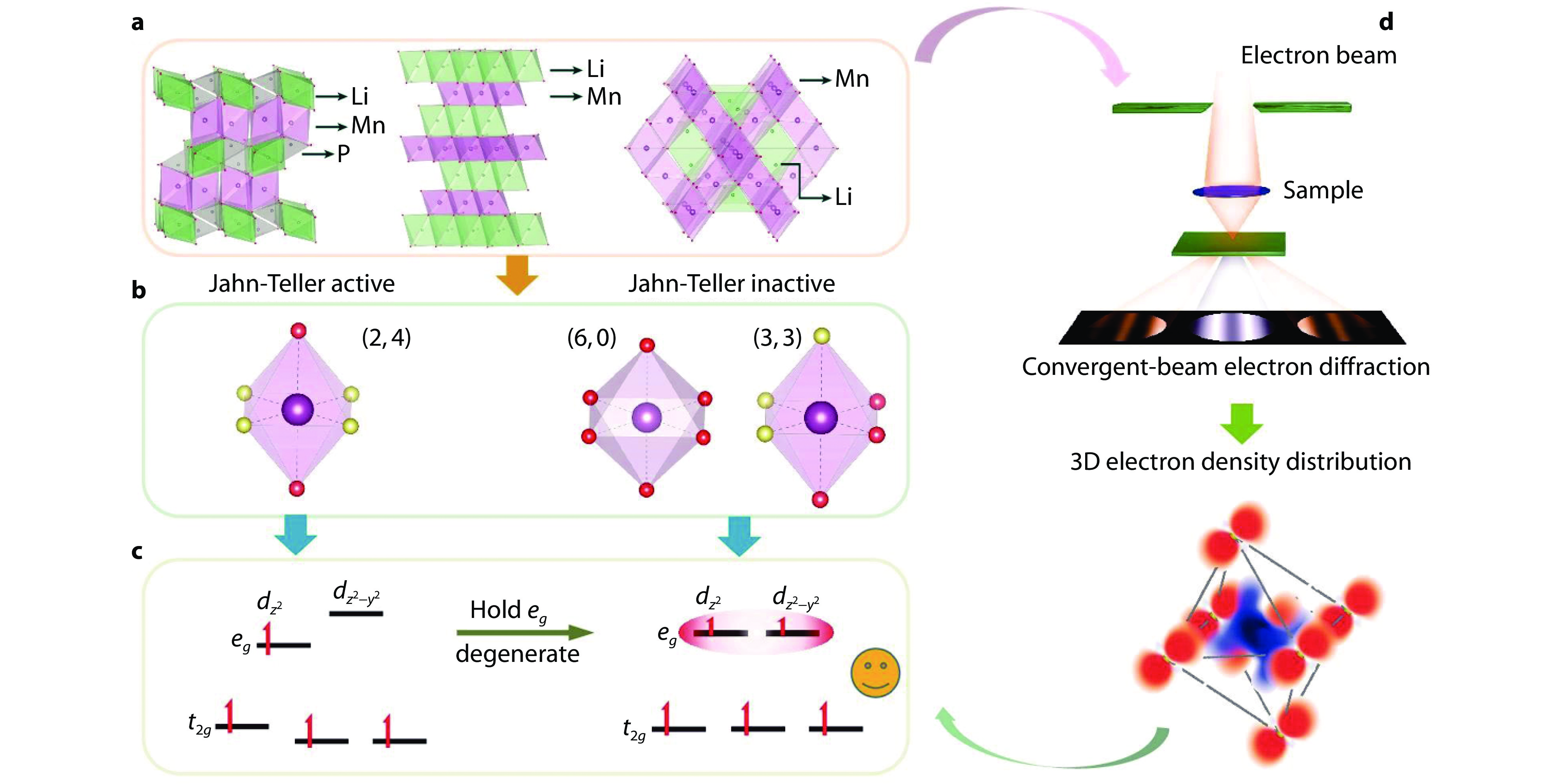
Figure 2.
Orbital strategy to control the Jahn–Teller effects (JTE). a Three common structure models (olivine, layered, spinel) of cathode materials for rechargeable Li-ion battery. b MnO6 octahedra with different symmetries. Ligands with the same color are identical and vice versa. The left octahedron is JT active, while the right two octahedra are JT inactive. c The orbital strategy to maintain the degeneracy of eg levels to suppress the JTE. d By combining convergent-beam electron diffraction and density functional theory (DFT) calculations, d-orbital occupancy can be quantified, and therefore the JTE can be detected [34]. Copyright 2024, Oxford University Press.
-
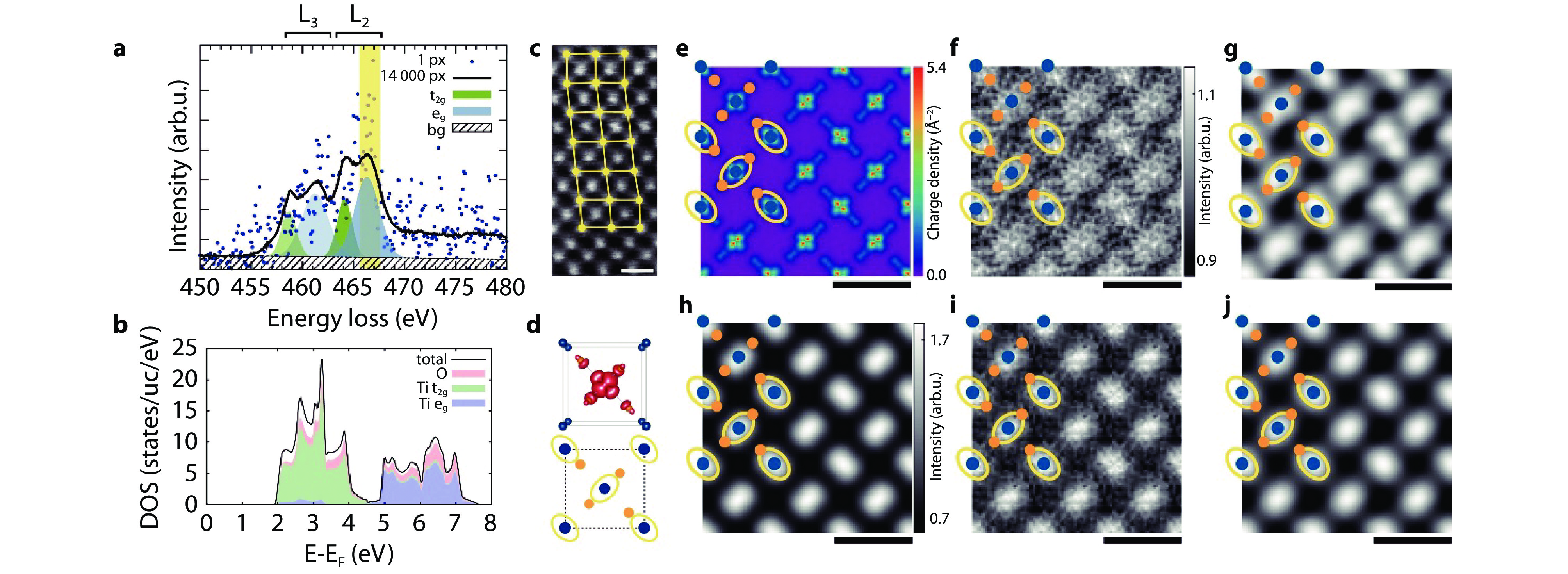
Figure 3.
Real-space mapping of electronic orbitals in Rutile with STEM-EELS. a Ti L2,3 edge extracted from a single pixel (dots), and averaged over all
14000 pixels (line) of the data set. The energy window used for the energy-filtered eg maps is highlighted in yellow. Gaussian least squares fits representing the individual shapes of the eg and t2g contributions are depicted in blue and green. b Projected DOS above the Fermi energy EF calculated by WIEN2k. c Dark field image acquired simultaneously with the spectrum image dataset. d Unit cell along the [0 0 1] direction used in the experiment with the summed three-dimensional charge density of the eg Wannier functions. Also shown are the projected positions of Ti (blue) and O (orange) atoms as well as yellow ellipses indicating the nearest O neighbours of each Ti (due to the projection, only two of the four nearest neighbours are visible). e Charge density for the unoccupied eg orbitals projected along the [0 0 1] crystallographic axis as calculated by WIEN2k. f Experimental energy-filtered map for the Ti L ionization edge for final states with eg character after unit-cell averaging. g Same as f, but after Gaussian smoothing. h Simulated energy-filtered map using the multislice algorithm and the mixed dynamic form factor approach after Gaussian blurring. i Same as h with added noise to better mimic the experimental conditions. j Same as i after Gaussian smoothing. All maps are replicated in a 3 × 3 raster for better visibility. Overlays show the projected positions of Ti and O atoms as well as yellow ellipses indicating the nearest O neighbours of each Ti. All scale bars indicate 5 Å[39]. Copyright 2017, Elsevier. -
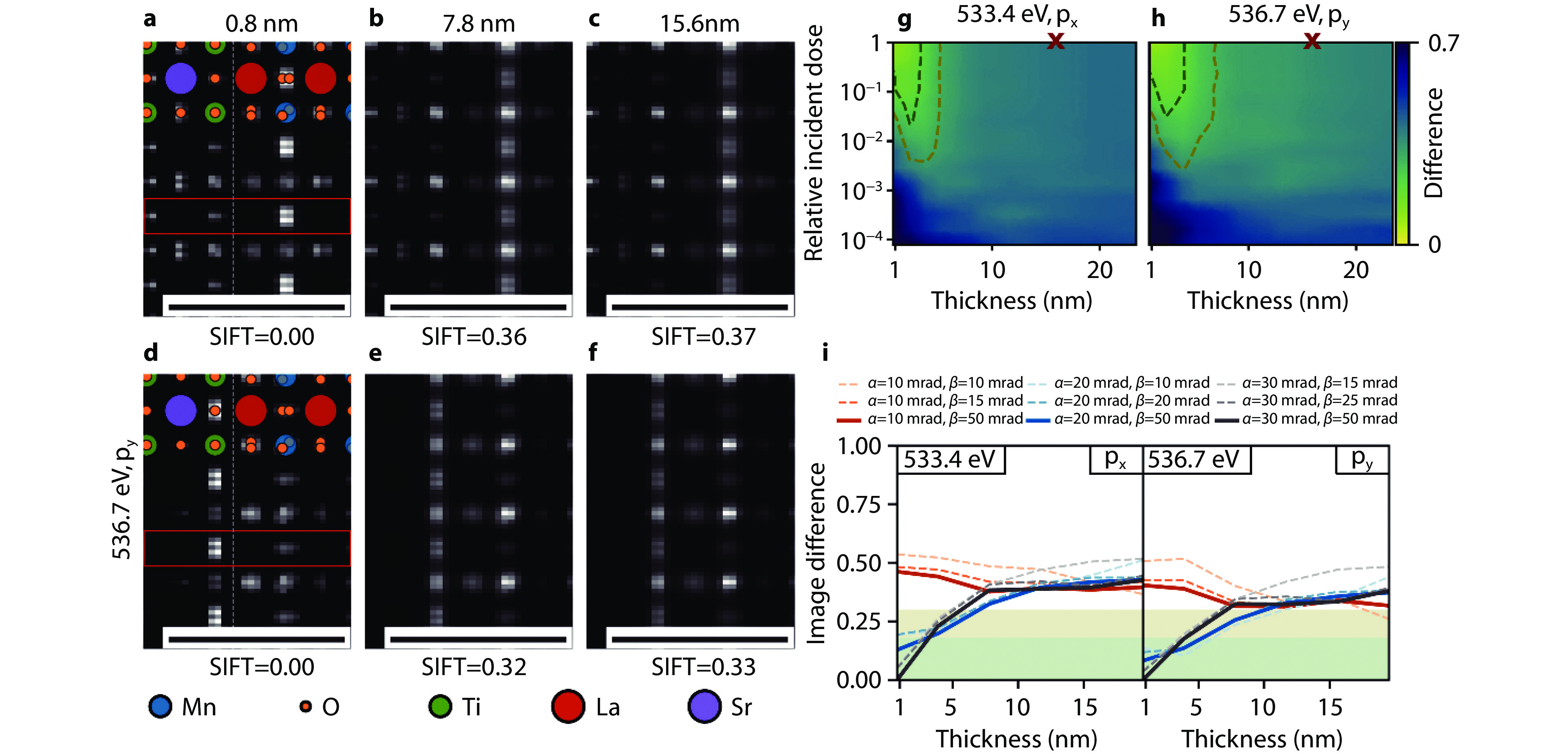
Figure 4.
Optimizing experimental parameters for orbital mapping in a heterostructure with EELS. a-f Simulated STEM spectrum images of the interface between STO and LMO in the [100] zone axis for both materials, the indicated sample thickness and energy loss with an infinite electron dose. All scale bars indicate 1 nm. g-h Image difference maps of the STO-LMO spectrum images for the indicated energy losses. The incident dose is measured relative to the image dose of 106 e−/nm2 for the image of a 15.6 nm thick sample, marked by the red x. The reference image in all cases is calculated with infinite electron dose. The dark green and olive contour lines indicate an image difference of 0.18 and 0.3, respectively. i STO-LMO SIFT image difference as a function of sample thickness for an acceleration voltage of 300 kV and several convergence and collection semi-angles α and β. All image differences are relative to the orbital map for α = 30 mrad and β = 50 mrad[46]. Copyright 2024, Elsevier.
-
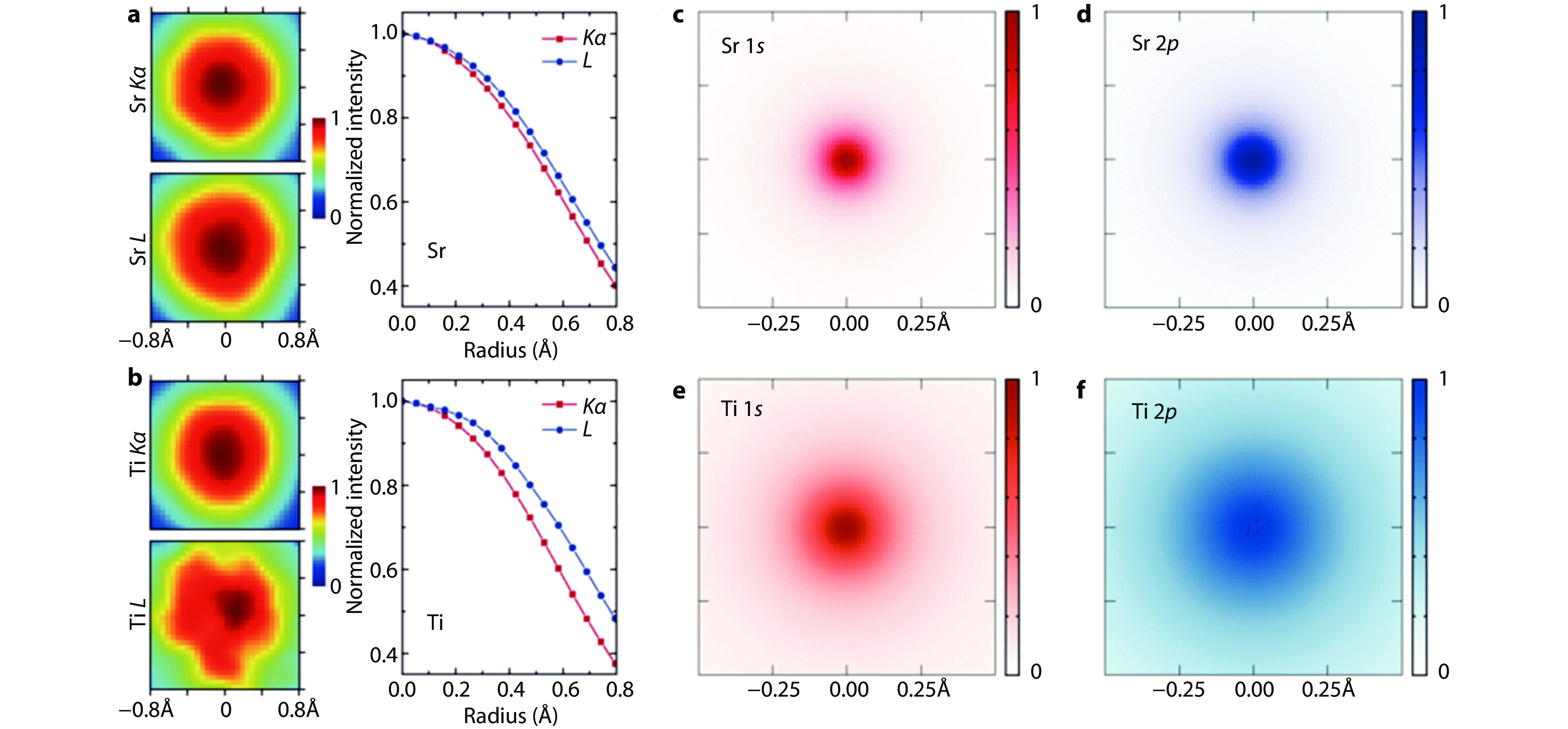
Figure 5.
Probing core-electron orbitals of Sr and Ti atoms in SrTiO3 with STEM-EDX. a Individual Sr Kα and L EDX maps from the Sr column of STO viewed along the [001] crystallographic direction. b Individual Ti Kα and L EDX maps from the Ti/O column of STO viewed along the same direction. The less circular shape of the Ti L map is due to a much lower signal-to-noise ratio in the data, showing that the Ti L signal still needs to be improved. For direct comparison, maps are background subtracted and normalized to their central intensity. Azimuthally averaged radial profiles are presented at right for better comparison. These maps constitute the cross-correlated average of data from approximately 450 identical atomic columns and all obtained simultaneously in a single experiment. c-f Experimentally observed projected excitation potentials for 1s and 2p orbitals of Sr and Ti, including the effects of atomic thermal vibrations and excitation broadening[48]. Copyright 2016, American Physical Society.
-
Figure 6.
High-resolution molecular orbital imaging with STM. a Naphthalocyanine on NaCl (2 ML) on Cu (111) measured using a Cu tip, a CO tip in constant-current mode, and a CO tip in constant-height mode. The PIR (1st row) is measured at V=−1.65 V and the NIR (2nd row) at V=0.60 V. b Calculated images at z0=5.0 Å of the HOMO (3rd row) and LUMO (4th row) with an s-wave tip, a p-wave tip, and a mixed tip. All images: 27 Å×27 Å[51]. Copyright 2011, American Physical Society.
-
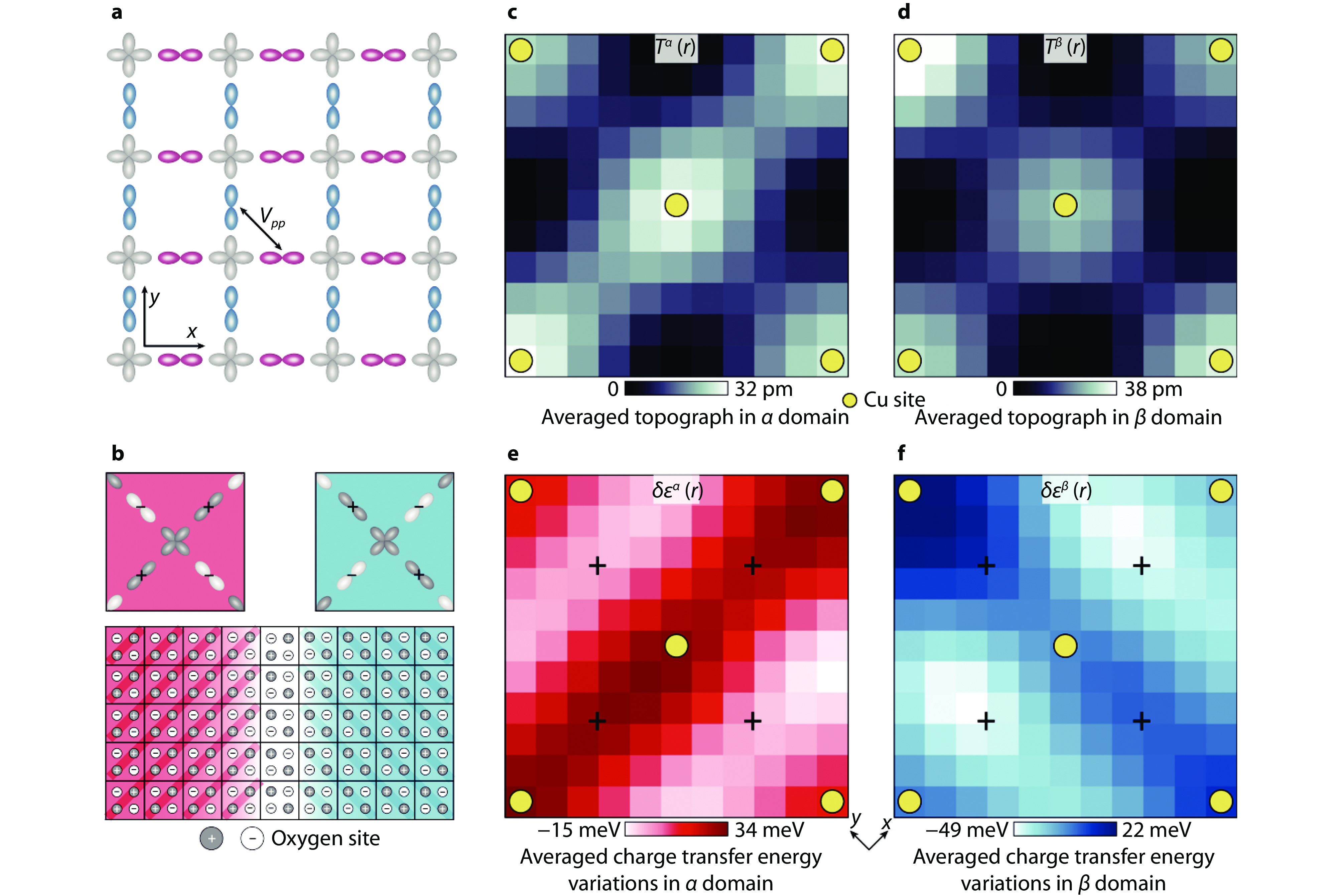
Figure 7.
Visualization of charge transfer energy and orbital ordering with STM. a Schematic of the relevant orbitals in the CuO2 plane depicting the crucial inter-oxygen-orbital Coulomb interaction Vpp. b Schematic of the charge-quadrupole two-level systems within the Ising-domain walls. The top panel shows the schematic of the charge distribution in the four intra-unit-cell oxygen sites consequent to the intra-unit-cell charge transfer symmetry breaking. The bottom panel shows a schematic of two orbitally ordered domains. The darker circles indicate oxygen sites with higher charge transfer energy ε. c Unit-cell-averaged structure of T(r) averaged over the regions where Nε > +5 meV. d Unit-cell-averaged structure of T(r) averaged over the regions where Nε < –5 meV. In both c and d, C4 symmetry is preserved. e Unit-cell-averaged structure of δε(r) averaged over the regions same as c. The charge transfer energy strongly breaks C4 symmetry about every Cu site (yellow dots), and consequently, there is an energy splitting of approximately 50 meV between the charge transfer energies at the two crystal-equivalent oxygen sites (indicated by the crosses). f Unit-cell-averaged structure of δε(r) averaged over the regions same as d. Virtually identical phenomena as in e, but rotated by 90°. The difference between e and f is the difference in the internal structure of the CuO2 unit cell in the two distinct Ising domains of orbital order[53]. Copyright 2024, Springer Nature.


 DownLoad:
DownLoad: